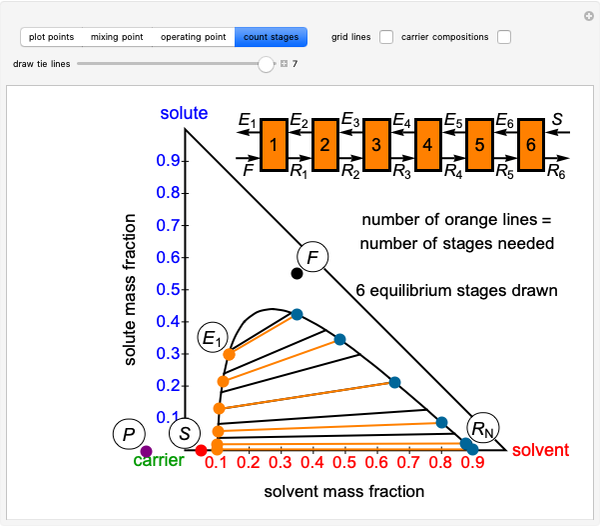
Add a Component to a Mixture with an Azeotrope

Requires a Wolfram Notebook System
Interact on desktop, mobile and cloud with the free Wolfram Player or other Wolfram Language products.
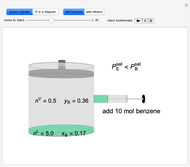
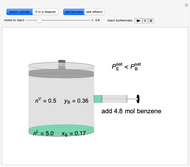
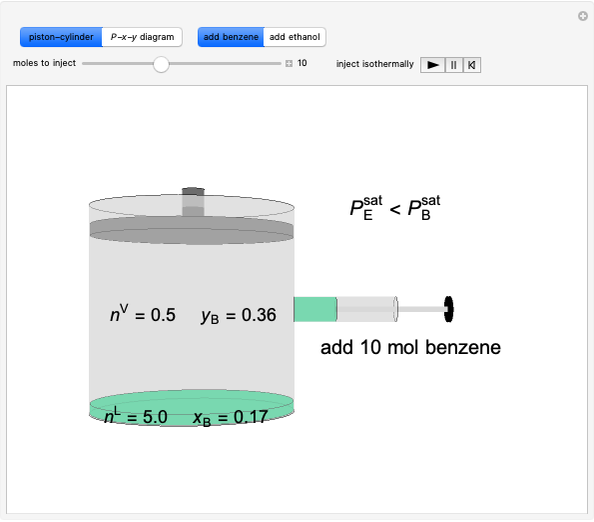
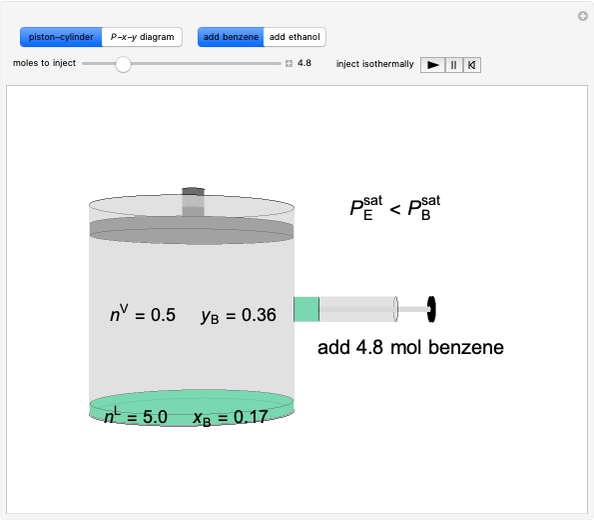
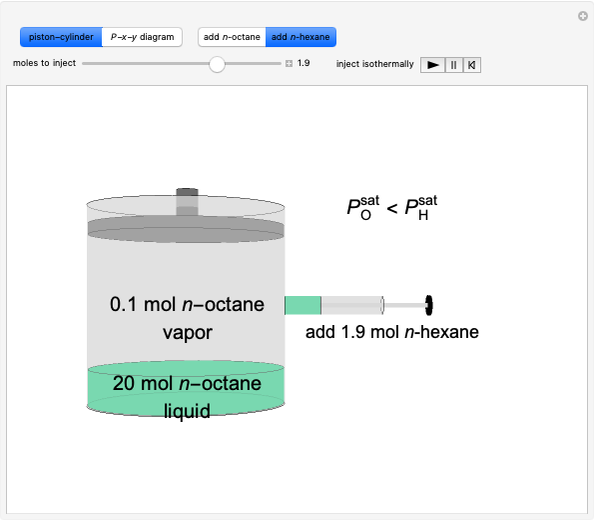
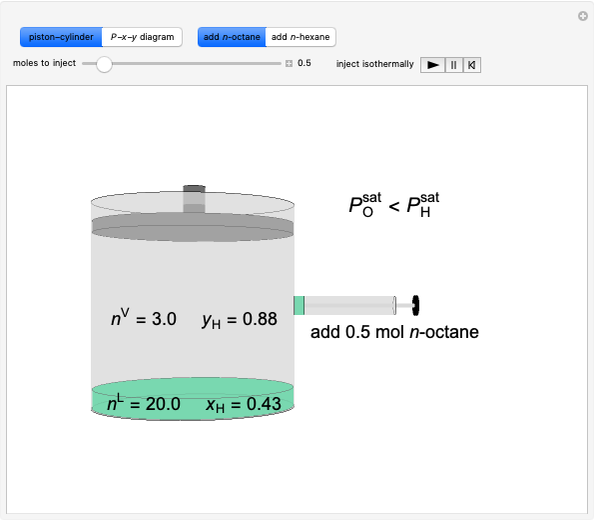
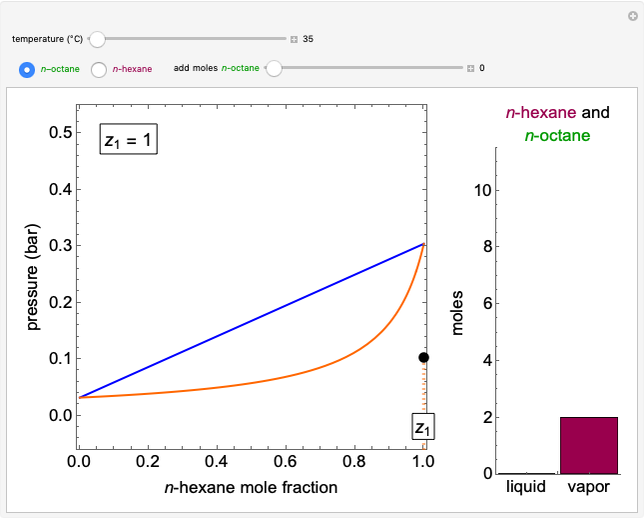
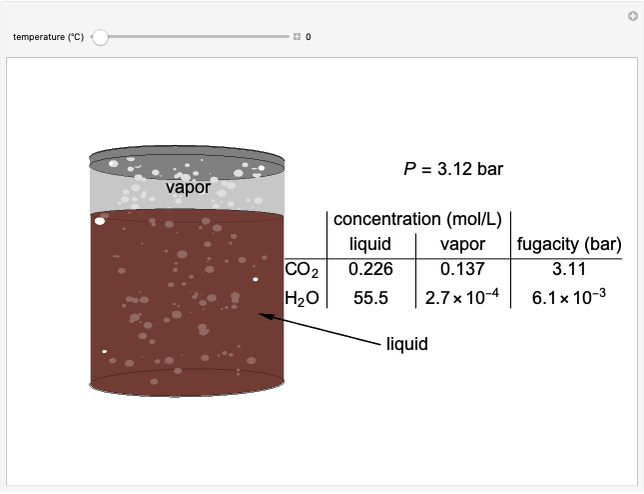
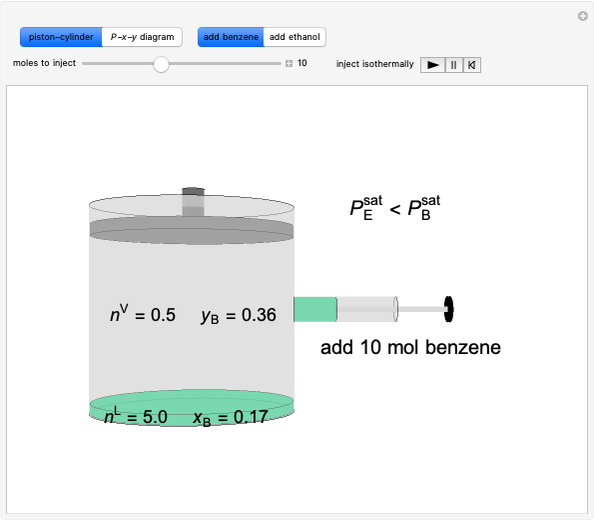
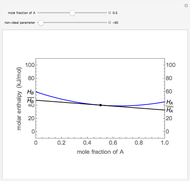
Benzene and ethanol, which have an azeotrope at a benzene mole fraction of 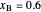 , are in vapor-liquid equilibrium (VLE) inside a cylinder fitted with a piston. Add either benzene or ethanol to the system by clicking the play button next to “inject isothermally”. The final state when the system returns to equilibrium after injection depends on which component is injected and how much is injected. The piston-cylinder shows the relative amounts of liquid and vapor, and the total moles in the liquid phase
, are in vapor-liquid equilibrium (VLE) inside a cylinder fitted with a piston. Add either benzene or ethanol to the system by clicking the play button next to “inject isothermally”. The final state when the system returns to equilibrium after injection depends on which component is injected and how much is injected. The piston-cylinder shows the relative amounts of liquid and vapor, and the total moles in the liquid phase 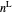 and in the vapor phase
and in the vapor phase 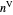 are displayed. The mole fraction of benzene in the liquid
are displayed. The mole fraction of benzene in the liquid 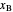 and the vapor
and the vapor 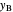 are also displayed. The
are also displayed. The 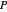 -
-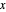 -
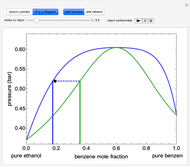
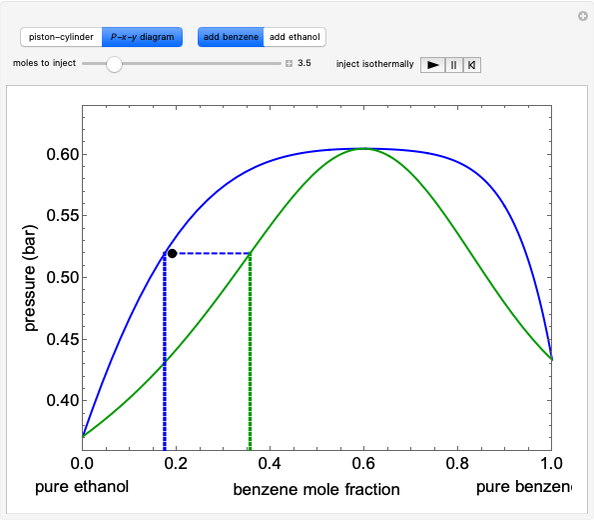
-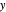 diagram shows the results of the mass balances and demonstrates why a given phase is the final state. To observe behavior for injecting a different number of moles or to inject the other component, click reset before clicking "inject isothermally" again.
diagram shows the results of the mass balances and demonstrates why a given phase is the final state. To observe behavior for injecting a different number of moles or to inject the other component, click reset before clicking "inject isothermally" again.
Contributed by: Rachael L. Baumann (December 2014)
Additional contributions by: John L. Falconer and Nick Bongiardina
(University of Colorado Boulder, Department of Chemical and Biological Engineering)
Open content licensed under CC BY-NC-SA
Snapshots
Details
The Antoine equation is used to calculate the saturation pressure 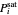 (bar) of each component:
(bar) of each component:
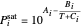 ,
,
where 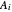 ,
, 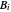 , and
, and 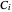 are the Antoine constants,
are the Antoine constants, 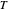 is temperature (°C), and the subscript
is temperature (°C), and the subscript 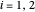 identifies the component (
identifies the component ( for benzene and
for benzene and 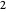 for ethanol).
for ethanol).
The fugacities 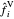 and
and 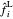 in the vapor and liquid phases (bar) are defined by:
in the vapor and liquid phases (bar) are defined by:
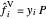 ,
,
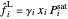 ,
,
where 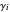 is the activity coefficient approximated by the two-parameter Margules equation,
is the activity coefficient approximated by the two-parameter Margules equation, 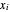 is the liquid mole fraction,
is the liquid mole fraction, 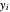 is the vapor mole fraction, and
is the vapor mole fraction, and 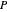 is the total pressure (bar).
is the total pressure (bar).
The fugacities are compared in order to determine the final phase of each component in the system. To have vapor-liquid equilibrium (VLE), the fugacities of each component must be equal in the liquid and vapor phases: 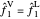 and
and 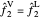 . After the addition of benzene or ethanol, components will either vaporize or condense in order to return to VLE. If the fugacity of the added component in the liquid phase is greater than that of the vapor phase, the components will vaporize:
. After the addition of benzene or ethanol, components will either vaporize or condense in order to return to VLE. If the fugacity of the added component in the liquid phase is greater than that of the vapor phase, the components will vaporize:
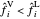 .
.
Conversely, if the fugacity of the vapor phase is greater, the components will condense to return to VLE:
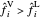 .
.
The total pressure is the sum of the partial pressures:
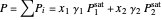 .
.
The activity coefficients are approximated by the two-parameter Margules equation:
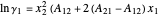 ,
,
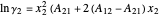 ,
,
where 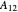 and
and 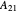 are the Margules parameters.
are the Margules parameters.
Finally:
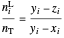 ,
,
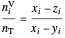 ,
,
where 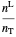 and
and 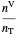 are the amounts of liquid and vapor in the system calculated from the lever rule and
are the amounts of liquid and vapor in the system calculated from the lever rule and 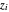 is the total mole fraction of a component.
is the total mole fraction of a component.
The screencast video at [1] explains how to use this Demonstration.
Reference
[1] Add a Component to a Mixture with an Azeotrope [Video]. (Dec 16, 2020) www.learncheme.com/simulations/separations/add-a-component-to-a-mixture-with-an-azeotrope.
Permanent Citation