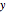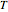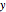Batch Distillation

Requires a Wolfram Notebook System
Interact on desktop, mobile and cloud with the free Wolfram Player or other Wolfram Language products.
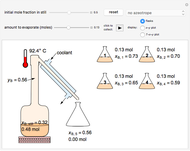
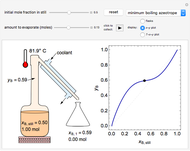
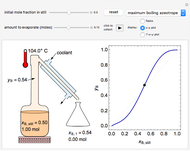
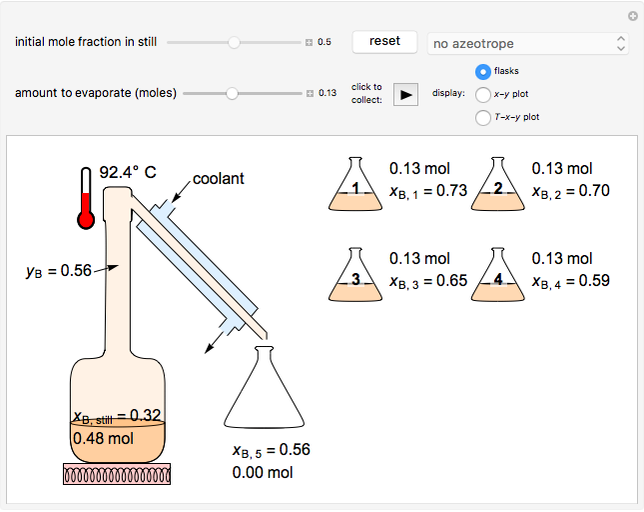
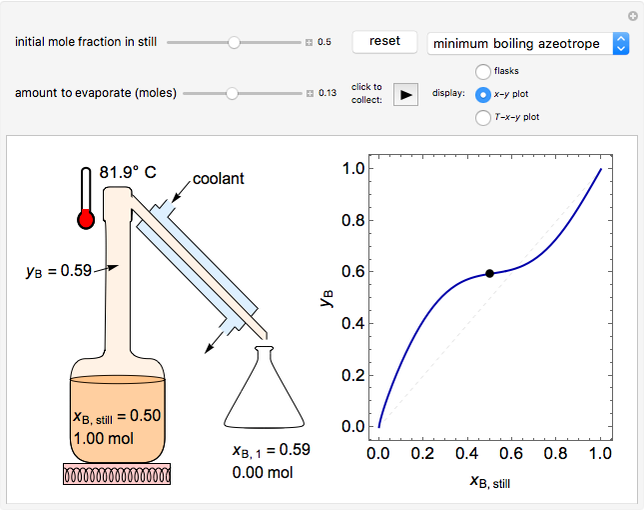
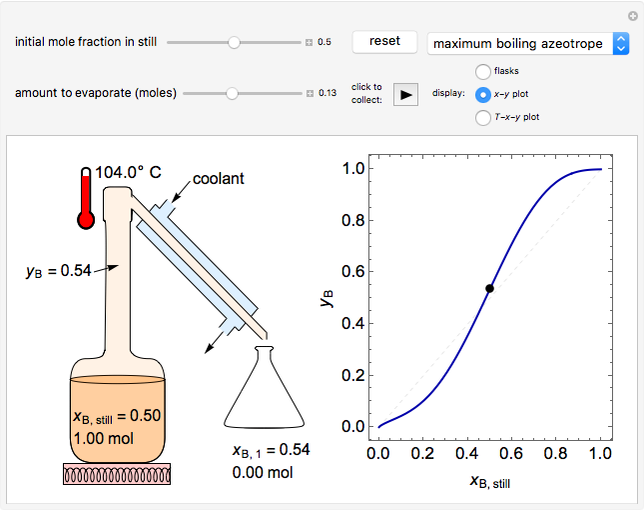
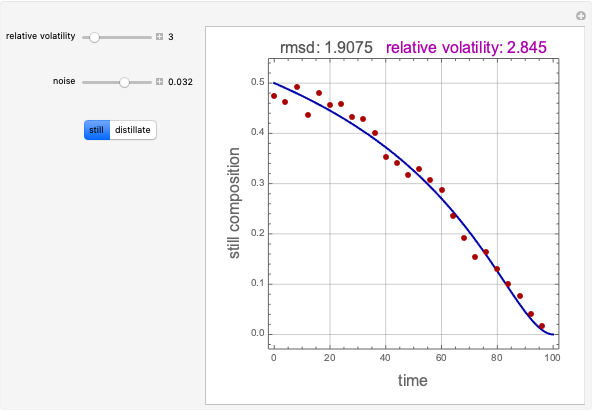
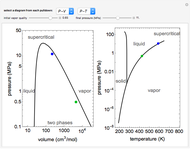
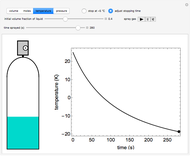
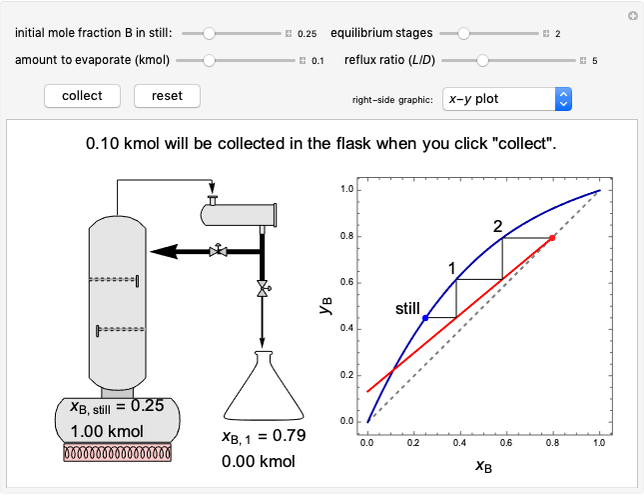
In this Demonstration, one mole of a binary mixture undergoes a batch distillation.
[more]
Contributed by: Neil Hendren (May 2019)
Additional Contributions by: John L. Falconer
(University of Colorado Boulder, Department of Chemical and Biological Engineering)
Open content licensed under CC BY-NC-SA
Details
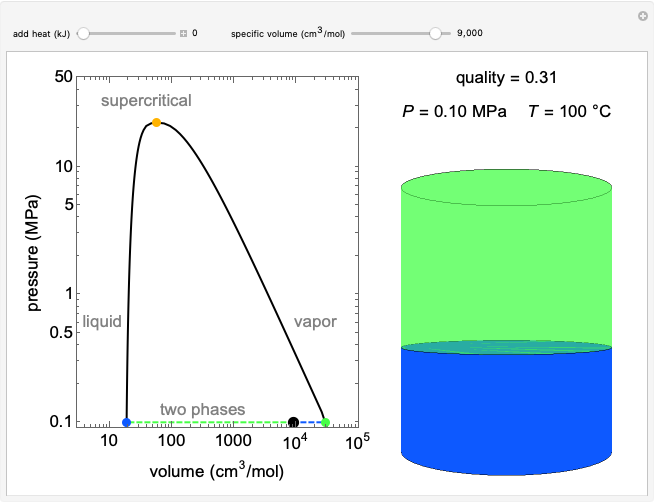
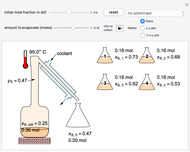
In batch distillation of a binary mixture, a fixed amount of the feed mixture 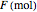 , with initial mole fraction
, with initial mole fraction 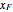 , evaporates into a distillate collection flask;
, evaporates into a distillate collection flask; 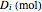 is the mass of distillate collected into flask
is the mass of distillate collected into flask 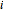 , and
, and 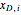 is the mole fraction within distillate flask
is the mole fraction within distillate flask 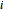 . This process is repeated to fill several distillate collection flasks. The overall mass balance is:
. This process is repeated to fill several distillate collection flasks. The overall mass balance is:
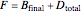 (1)
(1)
and the balance for the more volatile component is:
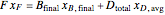 , (2)
, (2)
where 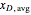 is the average mole fraction within the distillate collection flasks and
is the average mole fraction within the distillate collection flasks and 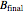 and
and 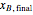 are the final mass and mole fraction within the bottom (boiler) vessel. The value of
are the final mass and mole fraction within the bottom (boiler) vessel. The value of 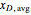 can be calculated as:
can be calculated as:
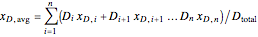 , (3)
, (3)
where the total number of collection stages is  (equivalent to the total number of distillate collection flasks).
(equivalent to the total number of distillate collection flasks).
Total distillate is given by:
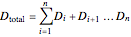 . (4)
. (4)
Because the saturated vapor is in thermodynamic equilibrium with the saturated liquid in the vessel (and there is only one equilibrium stage: the boiler/bottom vessel), the composition of the vapor 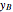 is a function of the composition in the bottom vessel. During evaporation, both compositions change with time, except when the composition of the liquid is an azeotrope. An equilibrium function is:
is a function of the composition in the bottom vessel. During evaporation, both compositions change with time, except when the composition of the liquid is an azeotrope. An equilibrium function is:
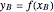 . (5)
. (5)
Numerical approximations such as Antoine's equation are often used to model 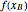 using experimental data. Other cases, such as constant relative volatility, may also be used to approximate this relationship.
using experimental data. Other cases, such as constant relative volatility, may also be used to approximate this relationship.
Composition of 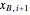 can be calculated by integration. Equation (5) must be substituted for
can be calculated by integration. Equation (5) must be substituted for 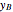 .
.
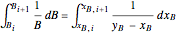 . (6)
. (6)
Finally, a mass balance can be used to solve for the distillate composition, 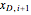 :
:
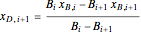 . (7)
. (7)
Reference
[1] P. C. Wankat, "Chapter 9: Batch Distillation," Separation Process Engineering: Includes Mass Transfer Analysis, 3rd ed., Upper Saddle River, NJ: Prentice Hall, 2012 pp. 329–347.
Snapshots
Permanent Citation