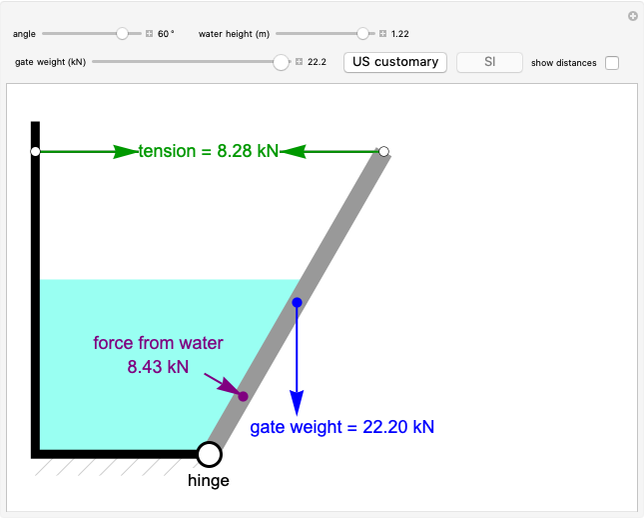
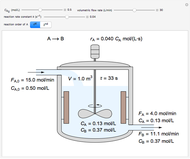
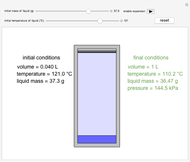
Construct a McCabe-Thiele Diagram for Distillation

Requires a Wolfram Notebook System
Interact on desktop, mobile and cloud with the free Wolfram Player or other Wolfram Language products.
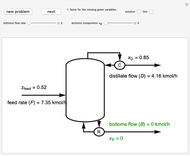
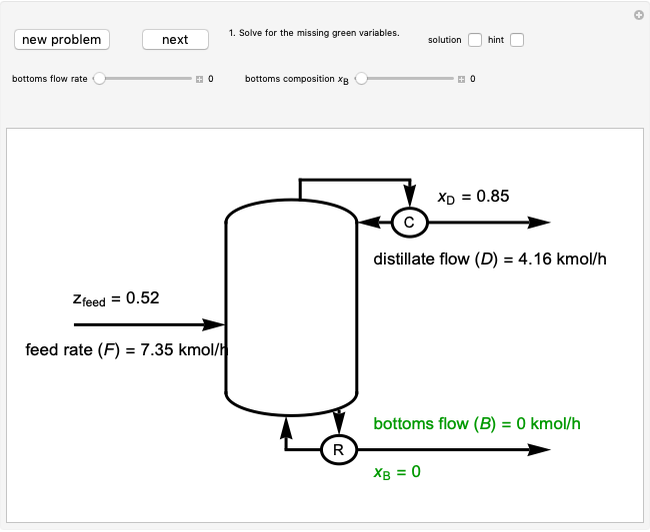
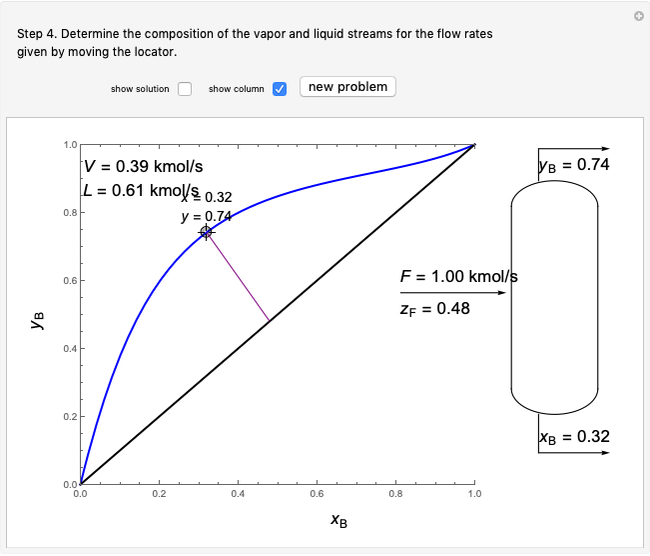
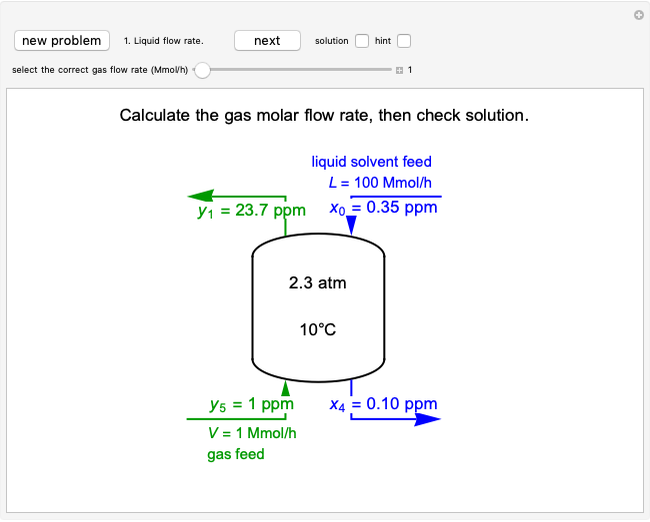
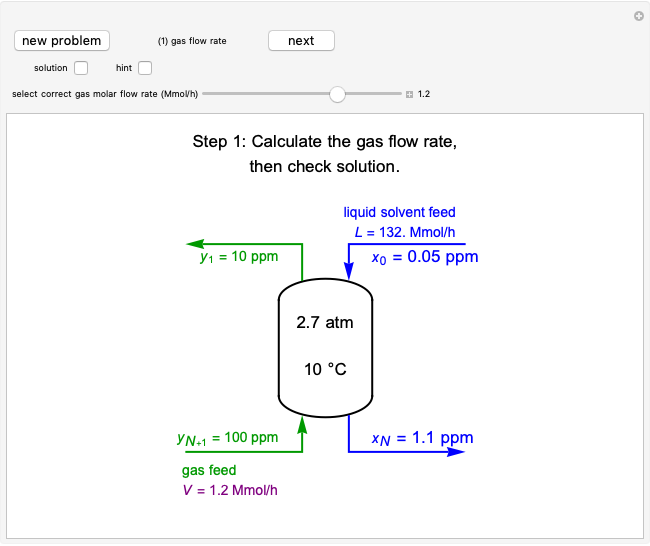
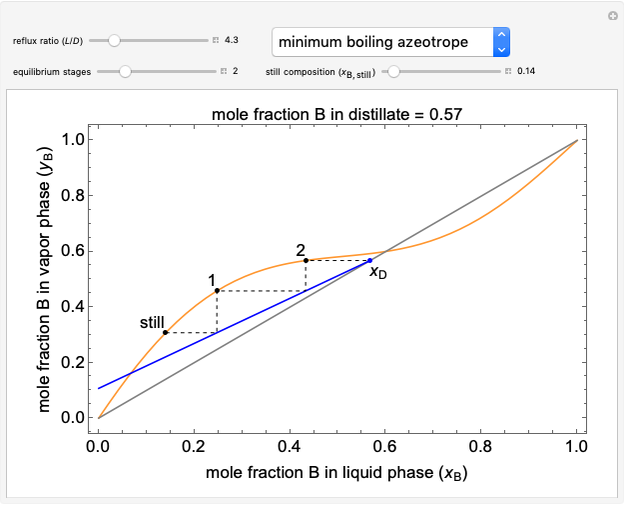
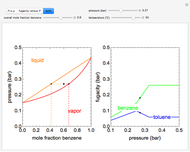
This Demonstration uses a step-by-step approach to calculate the number of stages for a specified distillation problem, using the McCabe–Thiele method. Click the "new problem" button to change the starting conditions. Check the "hint" box for a hint on how to proceed. After completing each step, check the "solution" box to see the correct solution and then click "next" for the next step. Once you check "solution," you cannot change your answer. Also, you cannot return to the previous step once you click "next."
Contributed by: Neil Hendren and Rachael L. Baumann (August 2022)
Additional contributions by: John L. Falconer
(University of Colorado Boulder, Department of Chemical and Biological Engineering)
Open content licensed under CC BY-NC-SA
Snapshots
Details
The McCabe–Thiele graphical solution method for binary distillation is used to determine the number of equilibrium stages needed to achieve a specified separation in a distillation column. This method assumes:
1. The distillation column is adiabatic.
2. Constant molar overflow (CMO), which means that for every mole of vapor condensed, one mole is vaporized on any equilibrium stage. This results in constant 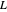 and
and 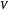 flow rates in the rectifying (upper) section, and constant
flow rates in the rectifying (upper) section, and constant 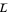 and
and 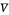 flow rates in the stripping (bottom) section. The symbols used for liquid and vapor flow rates are
flow rates in the stripping (bottom) section. The symbols used for liquid and vapor flow rates are 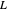 (liquid flow rate in the rectifying section),
(liquid flow rate in the rectifying section), 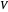 (vapor flow rate in the rectifying section),
(vapor flow rate in the rectifying section), 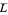 (liquid flow rate in the stripping section) and
(liquid flow rate in the stripping section) and 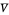 (vapor flow rate in the stripping section).
(vapor flow rate in the stripping section).
The constant molar overflow (CMO) assumption requires that:
2a. Specific heat of vaporization 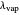 is the same for both components and thus independent of concentration.
is the same for both components and thus independent of concentration.
2b. Molar enthalpy changes ( ) between stages are small compared to latent heat changes, so
) between stages are small compared to latent heat changes, so
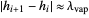 .
.
3. Heat of mixing is negligible.
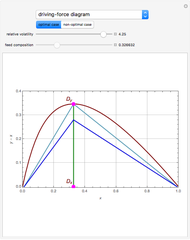
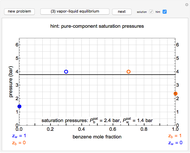
The equilibrium curve was calculated using the modified Raoult's law:
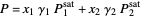 ,
,
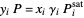 ,
,
where 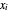 and
and 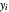 are the liquid and vapor mole fractions (
are the liquid and vapor mole fractions (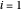 for the more volatile component,
for the more volatile component, 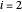 for the less volatile component),
for the less volatile component), 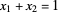 ,
, 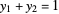 ,
,  is total pressure and
is total pressure and 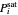 is the saturation pressure, which is calculated using the Antoine equation:
is the saturation pressure, which is calculated using the Antoine equation:
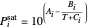 ,
,
where 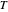 is temperature, and
is temperature, and 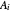 ,
, 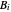 and
and 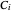 are Antoine constants.
are Antoine constants.
The activity coefficients 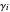 are calculated using the two-parameter Margules model:
are calculated using the two-parameter Margules model:
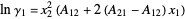 ,
,
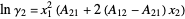 ,
,
where 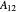 and
and 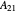 are the Margules parameters for a binary mixture. Typically, these are measured experimentally. In this Demonstration, Margules parameters are pseudorandom values.
are the Margules parameters for a binary mixture. Typically, these are measured experimentally. In this Demonstration, Margules parameters are pseudorandom values.
The method
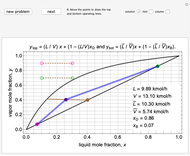
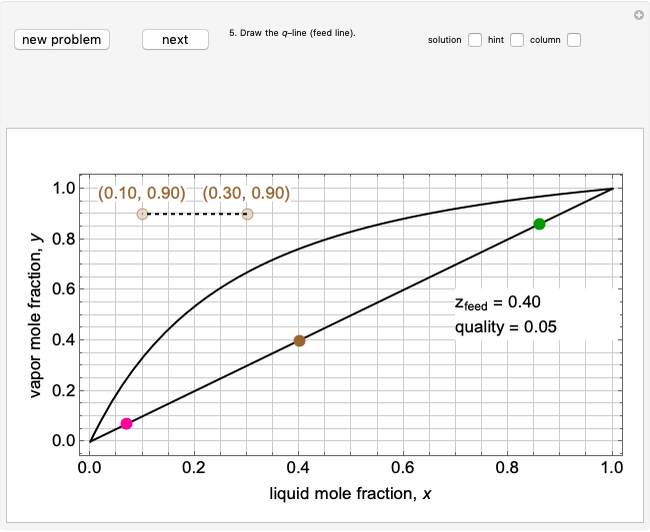
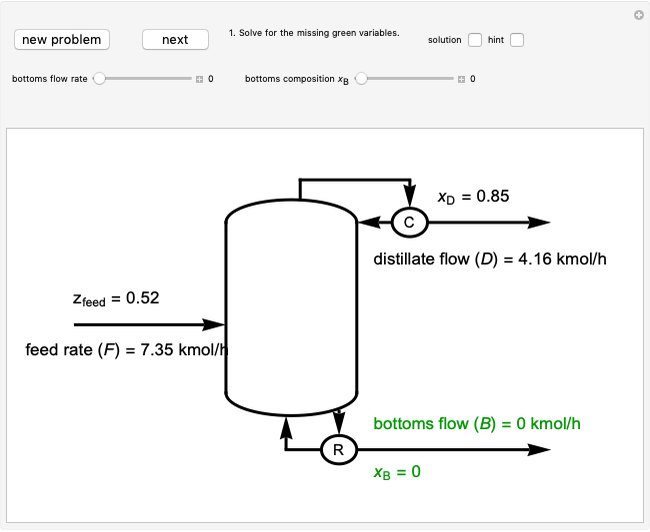
1. First the feed and rectifying operating lines are calculated. In this simulation
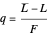 ,
,
where 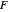 is the feed rate. Note that for subcooled liquids,
is the feed rate. Note that for subcooled liquids,  is greater than 1 and for superheated vapors,
is greater than 1 and for superheated vapors,  is less than 0. Equations for the feed line and the top and bottom operating lines are given below.
is less than 0. Equations for the feed line and the top and bottom operating lines are given below.
Feed line: 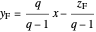 ,
,
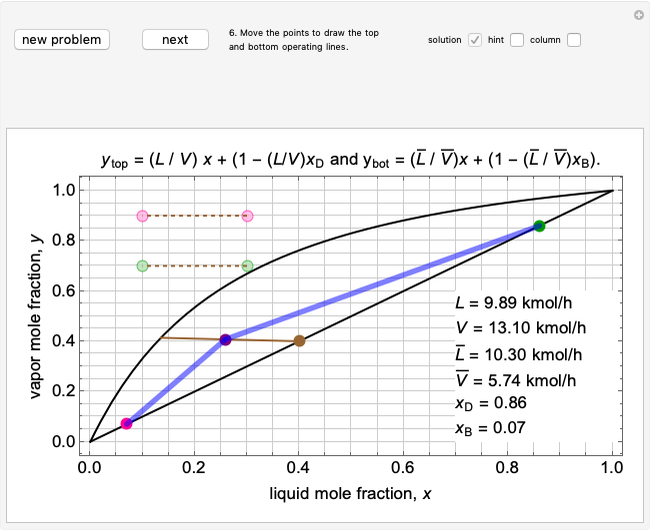
Top operating line: 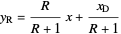 ,
,
Bottom operating line: 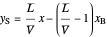 ,
,
where  is the quality of the feed
is the quality of the feed 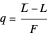 ,
, 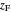 is the mole fraction of the more volatile component in the feed,
is the mole fraction of the more volatile component in the feed, 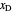 is the liquid mole fraction of the more volatile component in the distillate, and
is the liquid mole fraction of the more volatile component in the distillate, and 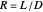 is the reflux ratio.
is the reflux ratio.
The intersection point of the top operating line, bottom operating line and feed line 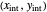 can be calculated:
can be calculated:
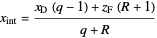 ,
,
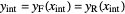 .
.
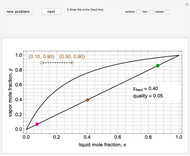
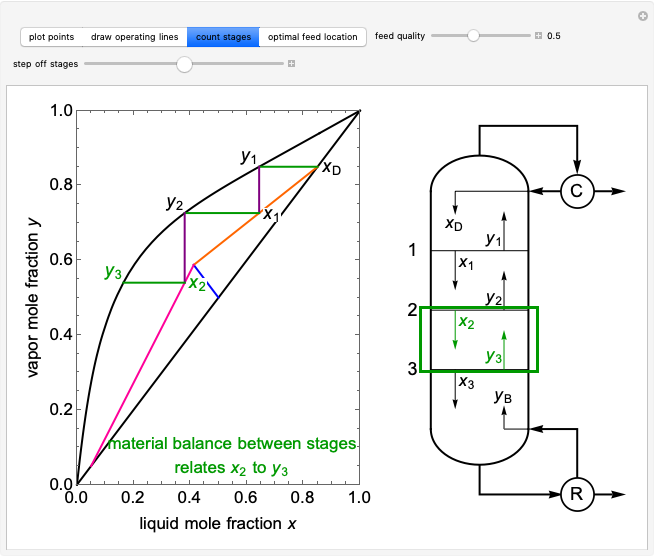
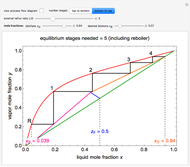
2. The number of stages is determined by "stair stepping" along the operating line, using the equilibrium line to determine the composition at each equilibrium stage. You can "stair step" from the bottom (reboiler stage) to the top (condenser stage) or from the top (condenser stage) to the bottom (reboiler stage). In this Demonstration, stairs are drawn from the top down.
3. The location of the feed stream is determined using the now fully specified graph. Locate the equilibrium stage that transitions from the top operating line to the bottom operating line. This is the stage where the feed stream must be put in order to get the specified distillate and bottoms compositions and flow rates.
View the screencast videos [1, 2] for a step-by-step explanation of the McCabe–Thiele method.
References
[1] G. Nicodemus. McCabe–Thiele Graphical Method Example Part 1 [Video]. (Jun 1, 2018) www.youtube.com/watch?v=Cv4KjY2BJTA.
[2] G. Nicodemus. McCabe-Thiele Graphical Method Example Part 2 [Video]. (Jun 1, 2018) www.youtube.com/watch?v=eIJk5uXmBRc.
Permanent Citation