Fugacities in an Ideal Binary Mixture

Requires a Wolfram Notebook System
Interact on desktop, mobile and cloud with the free Wolfram Player or other Wolfram Language products.
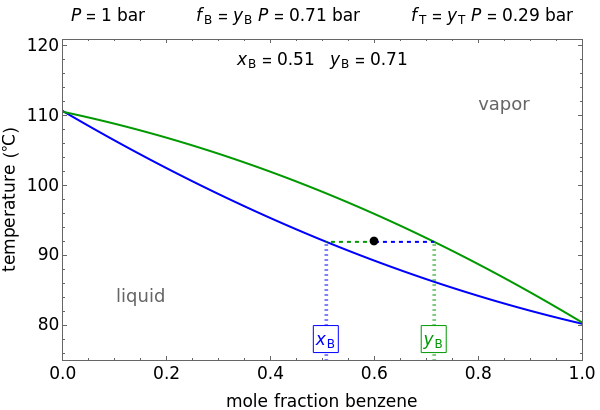
This Demonstration shows how the fugacities of benzene (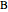 ) and toluene (
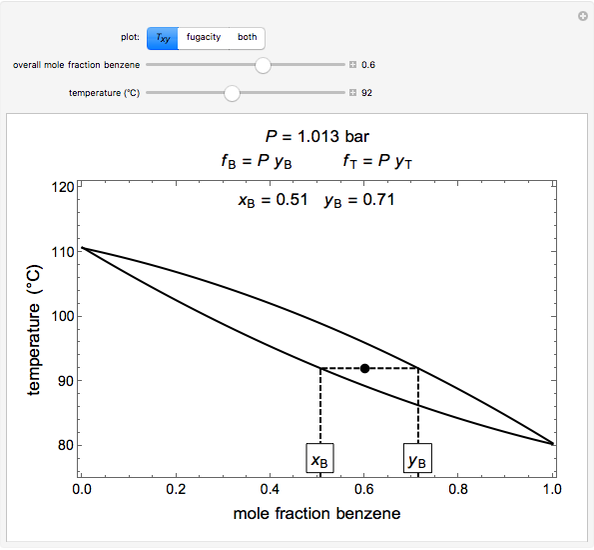
) and toluene ( ) change with temperature and molar composition at constant pressure. The liquid mixture is an ideal solution and the gas phase is ideal, so Raoult's law models vapor-liquid equilibrium. Use sliders to vary the temperature and overall mole fraction of benzene. Use buttons to view the temperature-composition diagram (
) change with temperature and molar composition at constant pressure. The liquid mixture is an ideal solution and the gas phase is ideal, so Raoult's law models vapor-liquid equilibrium. Use sliders to vary the temperature and overall mole fraction of benzene. Use buttons to view the temperature-composition diagram (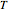 -
- -
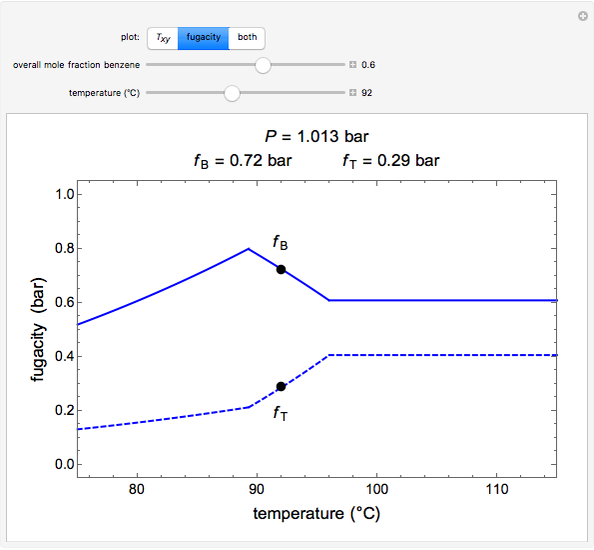
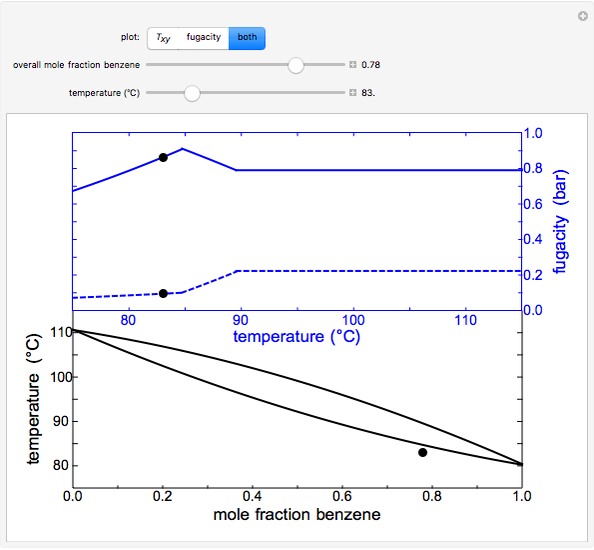
-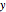 ), the fugacity-temperature plot or both plots at once.
), the fugacity-temperature plot or both plots at once.
Contributed by: Garrison J. Vigil and Rachael L. Baumann (July 2015)
Additional contributions by: John L. Falconer and Nick Bongiardina
(University of Colorado Boulder, Department of Chemical and Biological Engineering)
Open content licensed under CC BY-NC-SA
Snapshots
Details
The saturation pressures 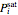 were calculated using the Antoine equation:
were calculated using the Antoine equation:
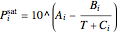 ,
,
where 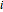 represents either benzene or toluene (
represents either benzene or toluene (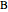 or
or  ),
), 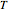 is temperature (°C), and
is temperature (°C), and 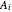 ,
, 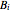 and
and 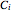 are Antoine constants.
are Antoine constants.
The fugacity 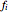 of a component depends on temperature and molar composition.
of a component depends on temperature and molar composition.
In the liquid region:
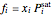 ,
,
where 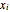 is the liquid mole fraction and both
is the liquid mole fraction and both 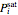 and
and 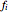 have units of bar.
have units of bar.
In the vapor region:
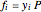 ,
,
where 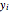 is the vapor molar composition and
is the vapor molar composition and 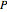 is the total pressure (bar).
is the total pressure (bar).
When vapor and liquid are in equilibrium (VLE):
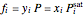 .
.
The screencast video at [1] explains how to use this Demonstration.
Reference
[1] Fugacities in an Ideal Binary Mixture [Video]. (Dec 1, 2016) www.colorado.edu/learncheme/thermodynamics/FugacitiesIdealBinaryMixture.xhtml.
Permanent Citation