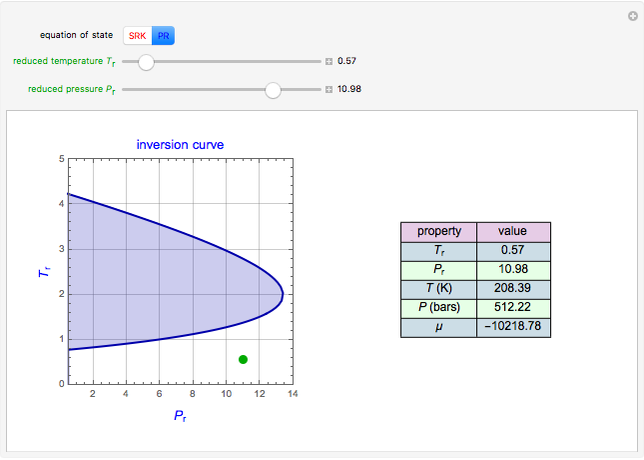
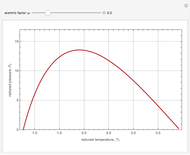
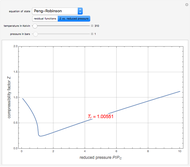
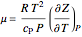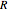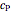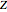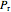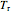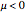Joule-Thomson Inversion Curves for Soave-Redlich-Kwong (SRK) and Peng-Robinson (PR) Equations of State
Initializing live version

Requires a Wolfram Notebook System
Interact on desktop, mobile and cloud with the free Wolfram Player or other Wolfram Language products.
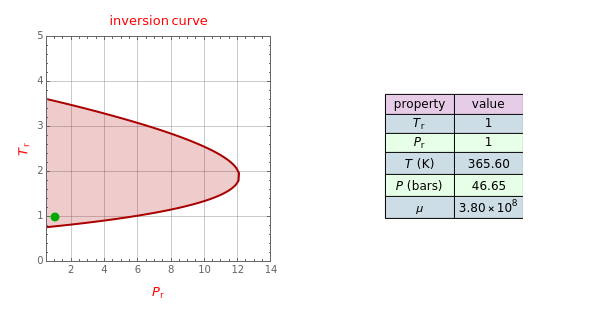
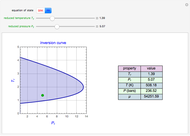
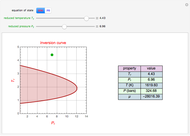
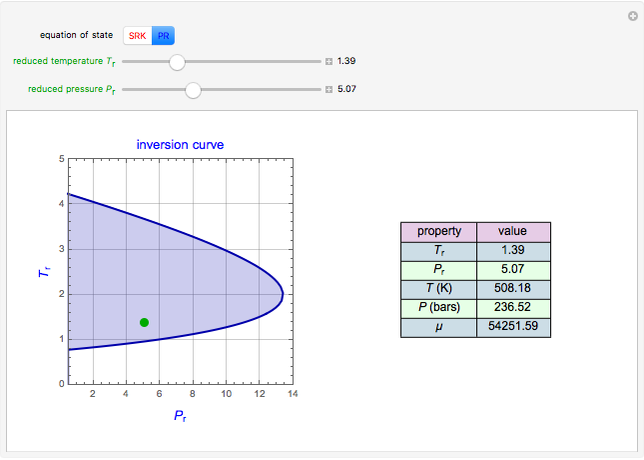
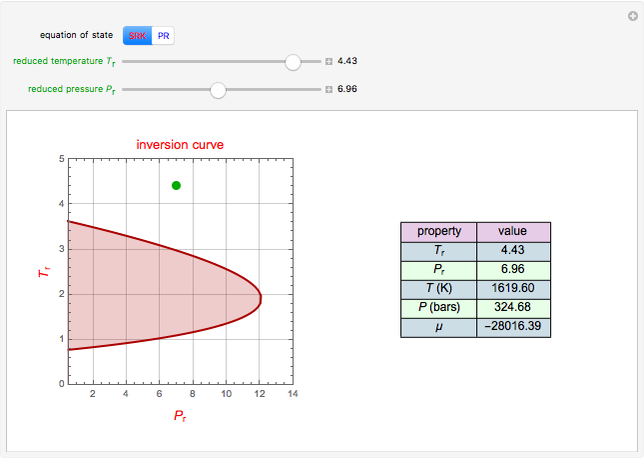
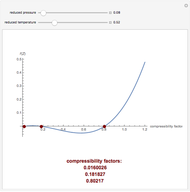
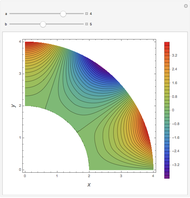
Throttling a real gas can sometimes result in a temperature decrease. If such is the case, the Joule–Thomson coefficient 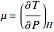 is positive. This coefficient can be written as:
is positive. This coefficient can be written as:
Contributed by: Housam Binous and Ahmed Bellagi (December 2016)
Open content licensed under CC BY-NC-SA
Snapshots
Details
Reference
[1] J. M. Smith, H. C. Van Ness and M. M. Abbott, Introduction to Chemical Engineering Thermodynamics, 7th ed., Boston: McGraw-Hill, 2005.
Permanent Citation