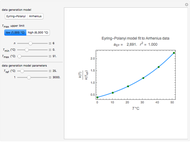
Michaelis-Menten Enzyme Kinetics of Bi-Bi Reactions

Requires a Wolfram Notebook System
Interact on desktop, mobile and cloud with the free Wolfram Player or other Wolfram Language products.
Enzyme kinetics describe the rate of chemical reactions catalyzed by enzymes and the effects of varying the reaction conditions. In an enzymatic reaction, specific molecules bind to an enzyme’s active site and are converted into products. Multiple-substrate reactions are more common than single-substrate reactions, accounting for over 60% of all enzymatic reactions. The following equation represents a bi-bi reaction in which  and
and  are substrates,
are substrates,  is the enzyme, and
is the enzyme, and  and
and  are the products:
are the products:
Contributed by: Bailey Schneider and Matthew Wick (April 2018)
Additional contributions by: Eitan Geva (University of Michigan)
Open content licensed under CC BY-NC-SA
Details
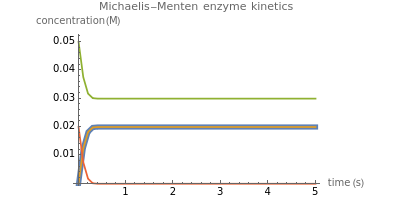
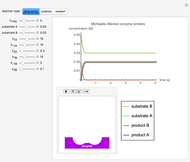
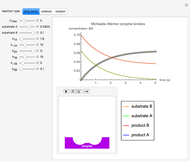
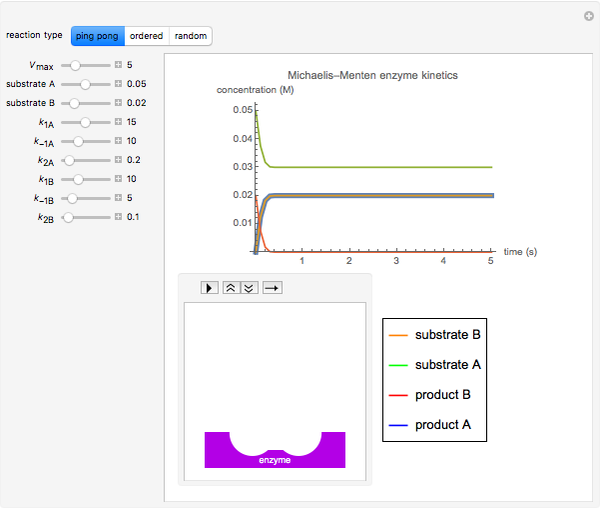
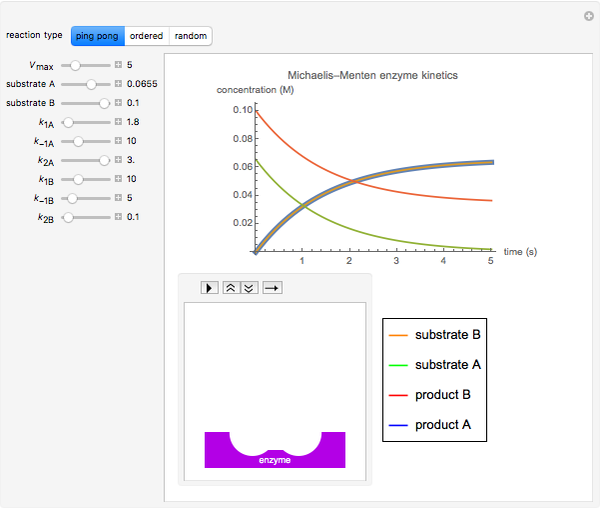
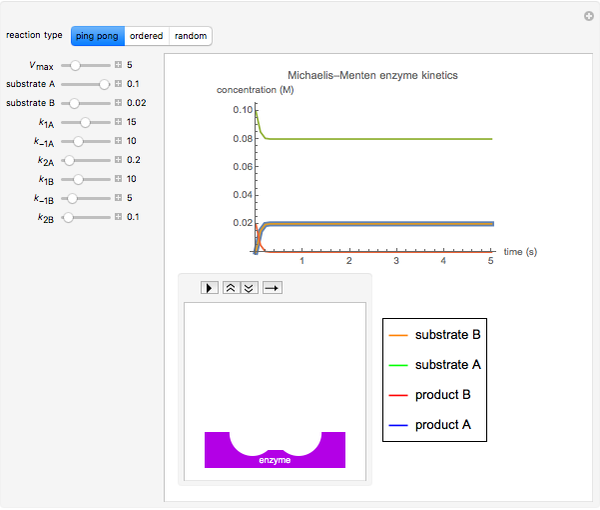
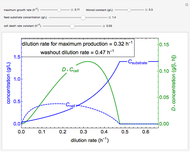
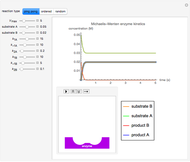
The Demonstration shows the formation of the intermediate complex derived from binding of the free enzyme and two other molecules (substrates  and
and  ). This model shows a general sequential reaction involving a single enzyme, two substrates and two products. The Michaelis–Menten enzyme kinetics graph shows a model for rate equations. Use the sliders to explore a variety of reaction conditions, including the initial concentrations of
). This model shows a general sequential reaction involving a single enzyme, two substrates and two products. The Michaelis–Menten enzyme kinetics graph shows a model for rate equations. Use the sliders to explore a variety of reaction conditions, including the initial concentrations of  and
and  , various rate constants and
, various rate constants and  . You can choose among three reaction types: ping pong, ordered and random. The lines of the graph represent a Michaelis–Menten steady-state approximation.
. You can choose among three reaction types: ping pong, ordered and random. The lines of the graph represent a Michaelis–Menten steady-state approximation.
References
[1] N. N. Ulusu, "Evolution of Enzyme Kinetic Mechanisms," Journal of Molecular Evolution, 80(5–6), 2015 pp. 251–257. doi:10.1007/s00239-015-9681-0.
[2] S. Tucek, "Choline Acetyltransferase and the Synthesis of Acetylcholine," in The Cholinergic Synapse, Handbook of Experimental Pharmacology, Vol. 86 (V. P. Whittaker, ed.), Berlin, Heidelberg: Springer, 1988 pp. 125–165. doi:10.1007/978-3-642-73220-1_ 7.
Submission from the Compute-to-Learn course at the University of Michigan.
Snapshots
Permanent Citation