Safe Operation of a Semibatch Reactor

Requires a Wolfram Notebook System
Interact on desktop, mobile and cloud with the free Wolfram Player or other Wolfram Language products.
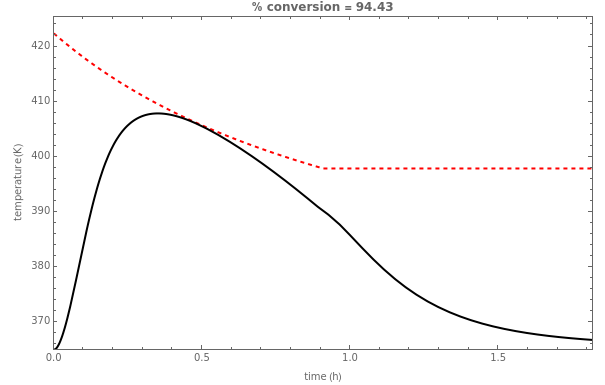
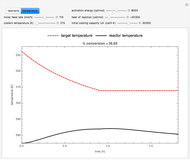
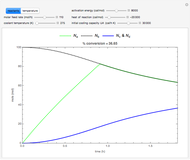
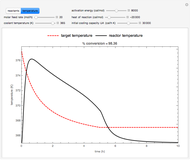
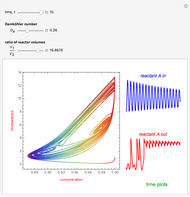
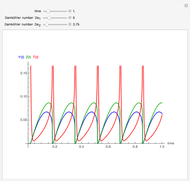
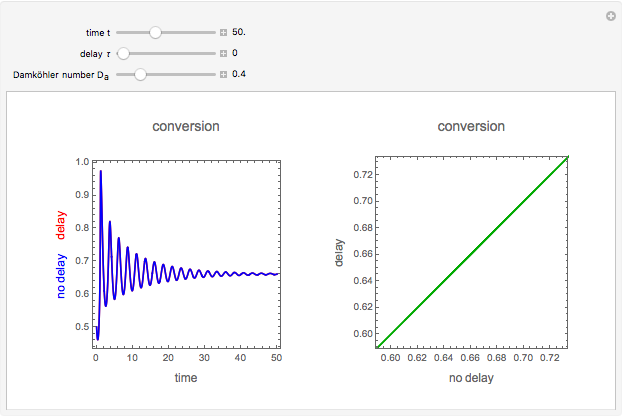
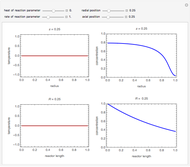
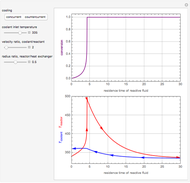
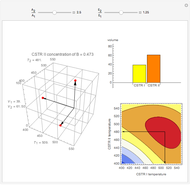
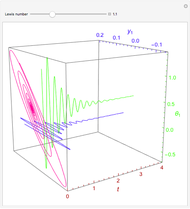
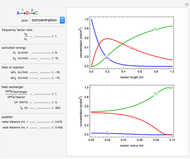
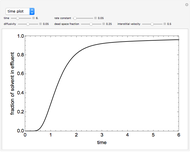
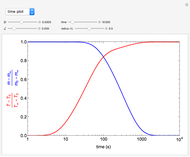
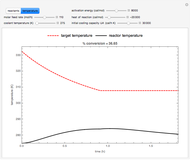
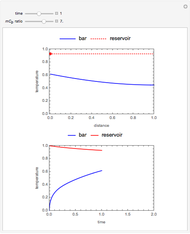
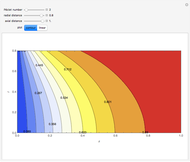
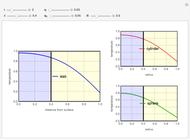
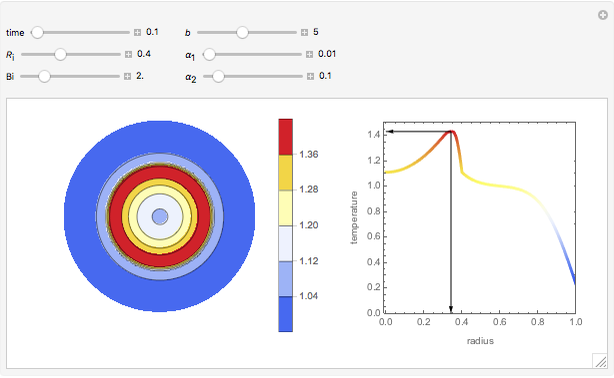
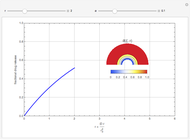
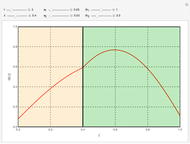
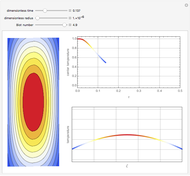
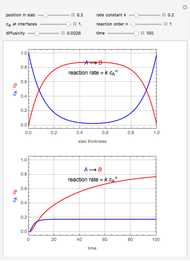
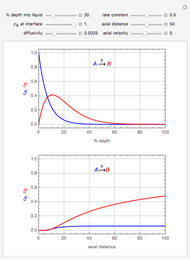
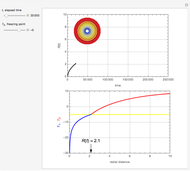
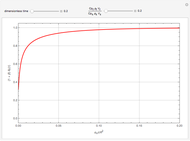
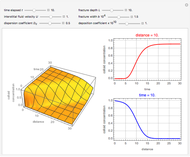
This Demonstration illustrates a method to operate a semibatch reactor to avoid thermal runaways.
[more]
Contributed by: Clay Gruesbeck (December 2017)
Open content licensed under CC BY-NC-SA
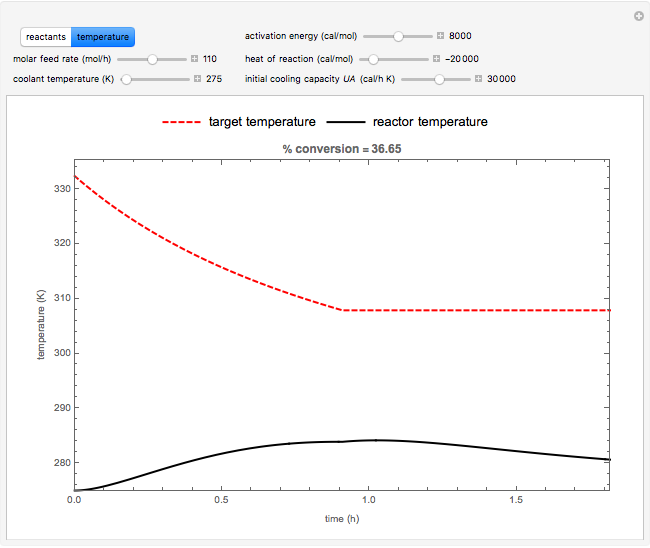
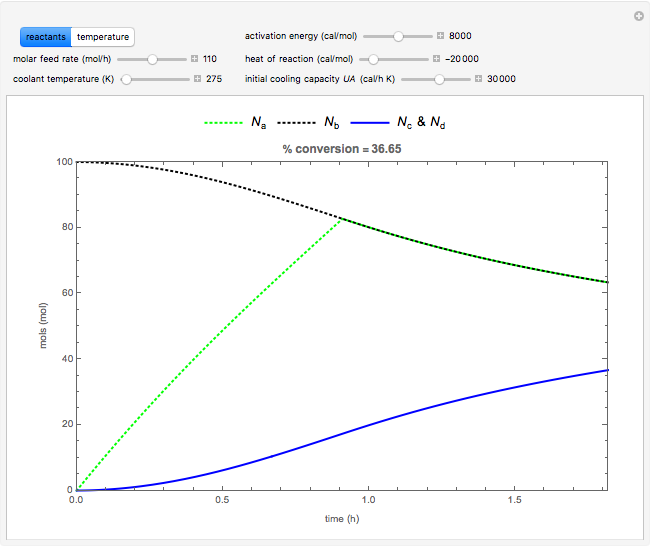
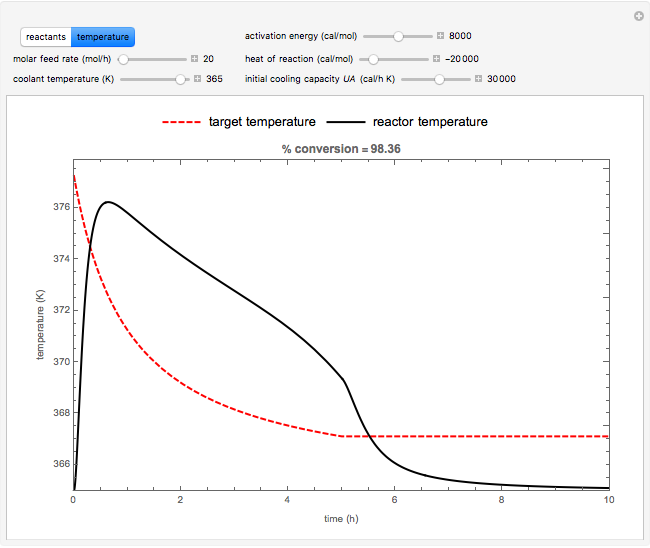
Snapshots
Details
The rate law for the reaction is:
The reactant and product concentrations are:
These are the material balances:
The energy balance is:
Initial conditions for these differential equations are:
Reference
[1] K. R. Westerterp, M. Lewak, and E. J. Molga, "Boundary Diagrams Safety Criterion for Liquid Phase Homogeneous Semibatch Reactors," Industrial and Engineering Chemistry Research, 53(14), 2014 pp 5778–5791. doi:10.1021/ie500028u.
Permanent Citation
"Safe Operation of a Semibatch Reactor"
http://demonstrations.wolfram.com/SafeOperationOfASemibatchReactor/
Wolfram Demonstrations Project
Published: December 14 2017