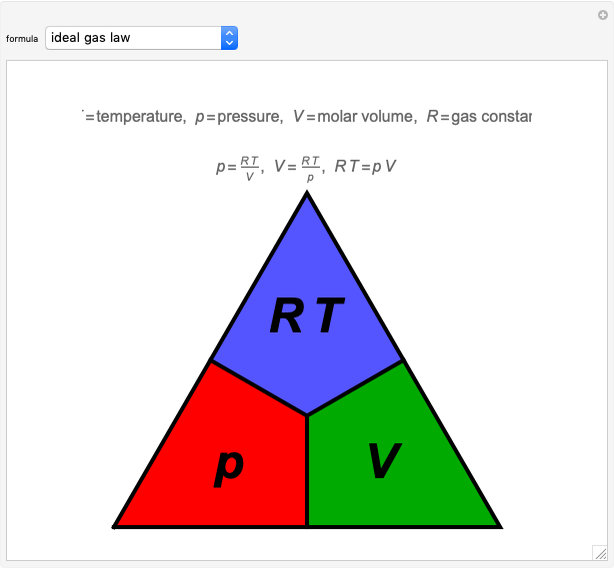
Diagrammatic Representations of Scientific Formulas

Requires a Wolfram Notebook System
Interact on desktop, mobile and cloud with the free Wolfram Player or other Wolfram Language products.
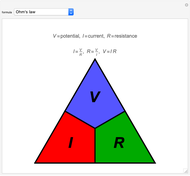
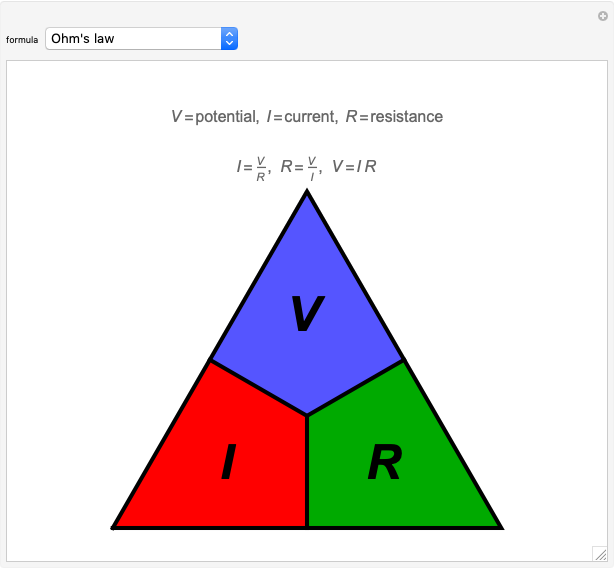
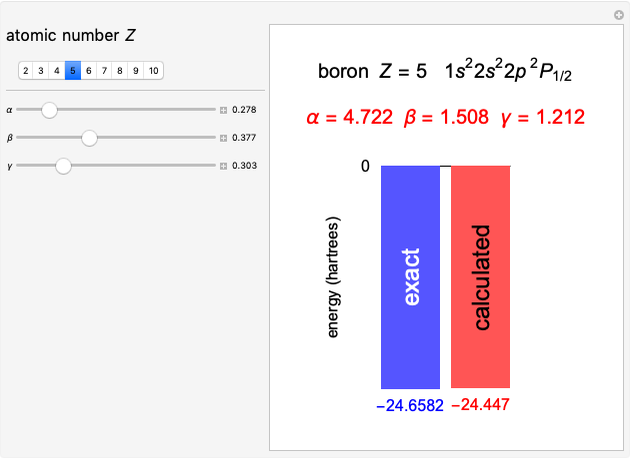
Ohm's law for electrical circuits can be read off a triangular diagram formed from the variables  ,
,  and
and  :
:
Contributed by: S. M. Blinder (June 2020)
Open content licensed under CC BY-NC-SA
Snapshots
Details
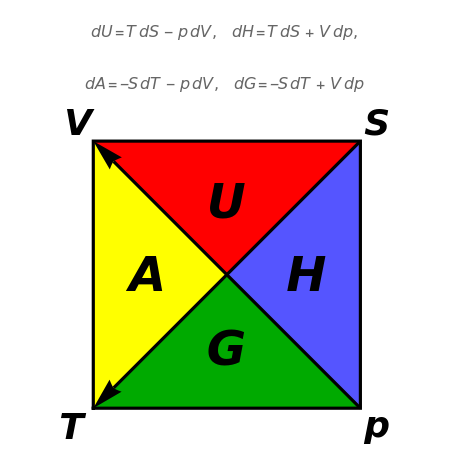
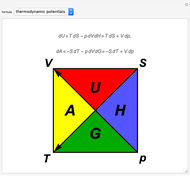
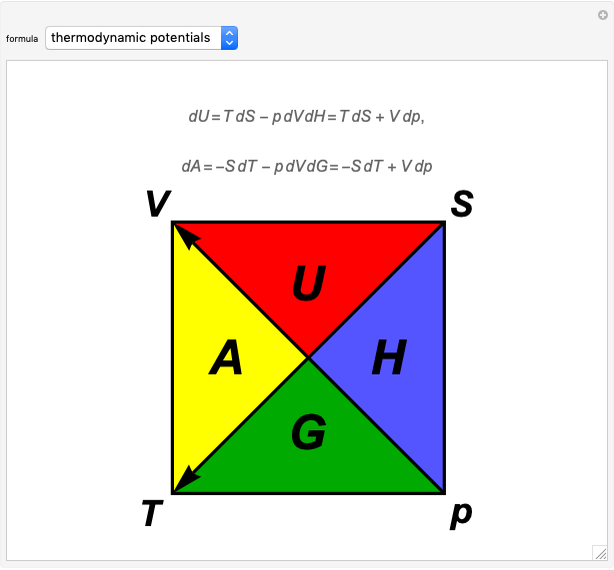
The thermodynamic potentials for a one-component system are energy  , enthalpy
, enthalpy  , Helmholtz free energy
, Helmholtz free energy  and Gibbs free energy
and Gibbs free energy  , each of which is a natural function of two state variables from among pressure
, each of which is a natural function of two state variables from among pressure  , volume
, volume  , temperature
, temperature  and entropy
and entropy  . The diagram identifies this functional dependence by the two vertices of the triangles for each of the thermodynamic functions. The fundamental differential relations for the potentials are given by
. The diagram identifies this functional dependence by the two vertices of the triangles for each of the thermodynamic functions. The fundamental differential relations for the potentials are given by
 ,
,
 ,
,
 ,
,
 .
.
The minus signs in these formulas can be deduced from the directions of the two diagonal arrows in the diagram. The Euler reciprocity conditions for each of the potentials, since they are exact differentials, give the four Maxwell relations:
 ,
,
 ,
,
 ,
,
 ,
,
which can also be read off the diagram.
References
[1] Ohm Law. "Ohm's Law Triangle." (Apr 29, 2020) ohmlaw.com/ohms-law-triangle.
[2] Wikipedia. "Thermodynamic Square." (Apr 29, 2020) www.eoht.info/page/Thermodynamic+square.
Permanent Citation