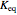Multiple Tubular Reactors with Interstage Cooling

Requires a Wolfram Notebook System
Interact on desktop, mobile and cloud with the free Wolfram Player or other Wolfram Language products.
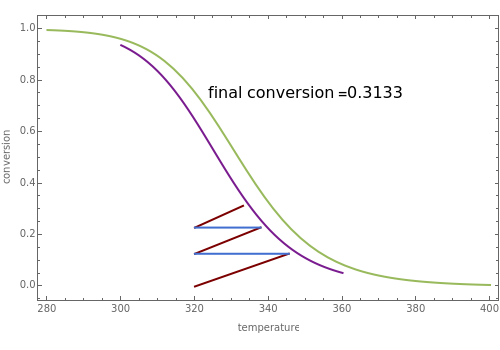
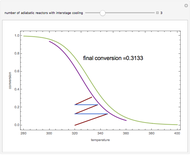
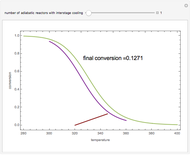
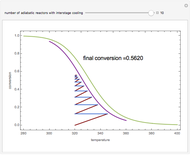
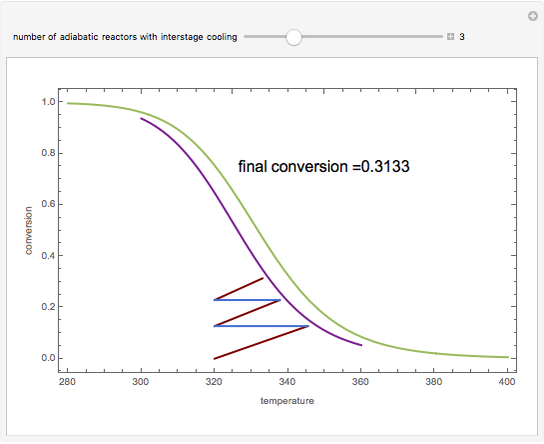
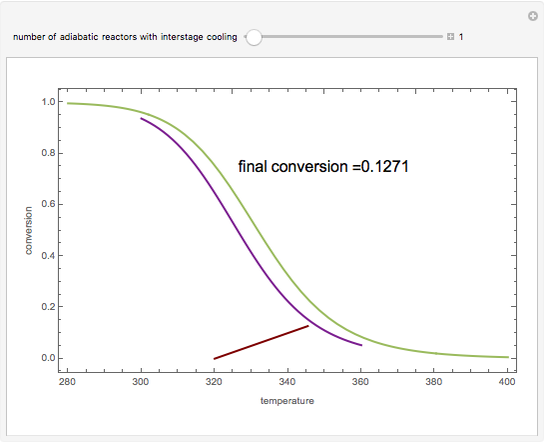
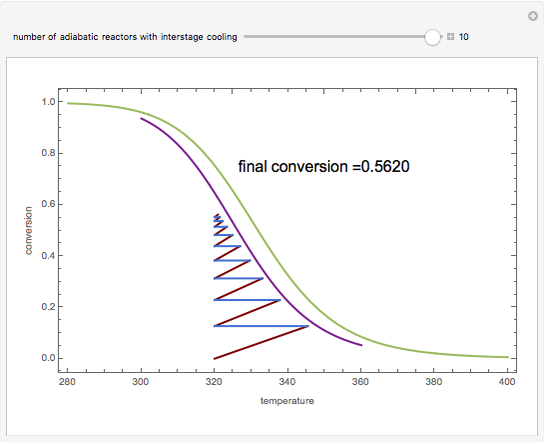
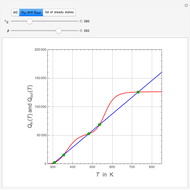
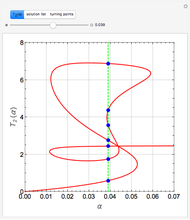
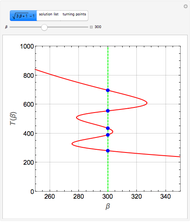
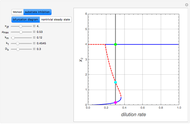
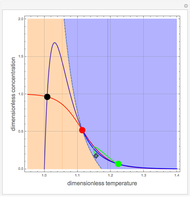
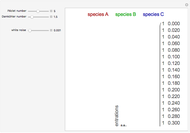
Low conversion is sometimes overcome using several adiabatic reactors with interstage cooling. Several reactors, with heat exchangers for cooling between them, are run up to near the maximum reaction rate until satisfactory conversion is attained. Methanol synthesis from syngas is a typical example where this technique is used:  . This reaction is reversible at 250°C (the temperature where commercial reactors are operated). It is common to use interstage cooling to achieve high conversion and to extract reaction heat.
. This reaction is reversible at 250°C (the temperature where commercial reactors are operated). It is common to use interstage cooling to achieve high conversion and to extract reaction heat.
Contributed by: Housam Binous (March 2011)
Open content licensed under CC BY-NC-SA
Snapshots
Details
L. D. Schmidt, The Engineering of Chemical Reactions, Oxford: Oxford University Press, 1998.
Permanent Citation