Potentiometric and Colorimetric Titration

Requires a Wolfram Notebook System
Interact on desktop, mobile and cloud with the free Wolfram Player or other Wolfram Language products.
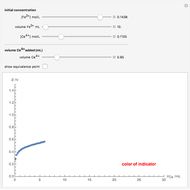
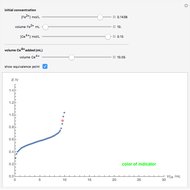
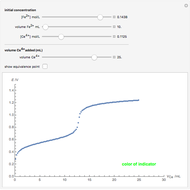
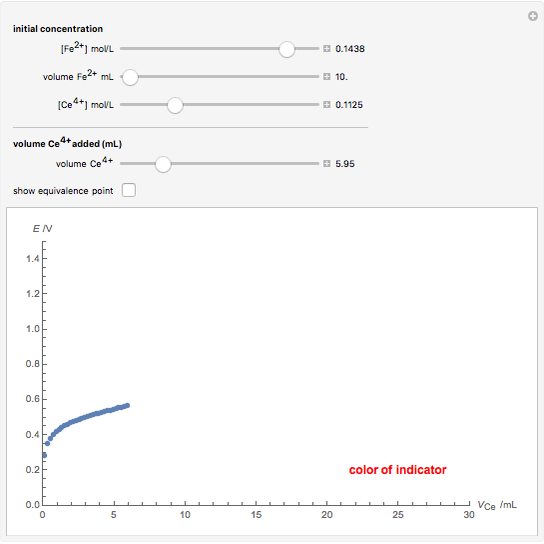
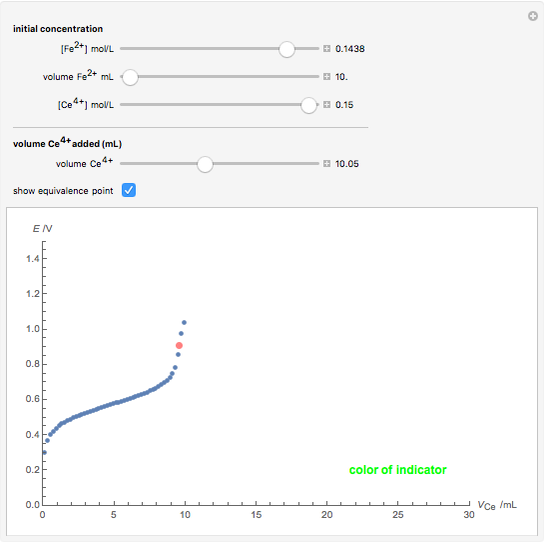
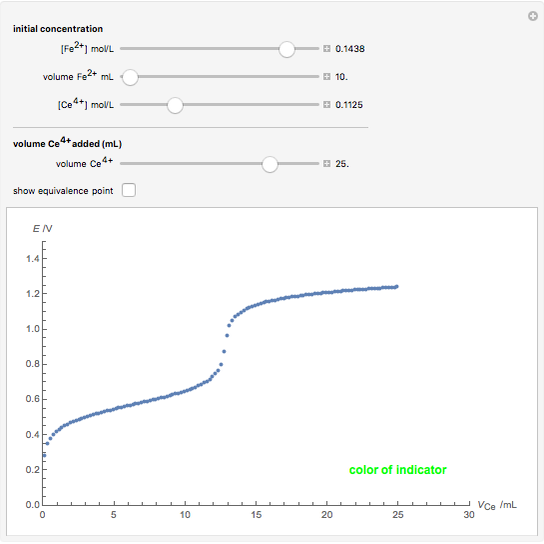
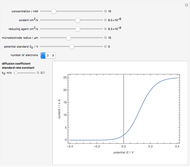
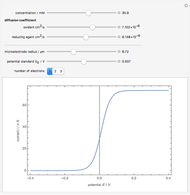
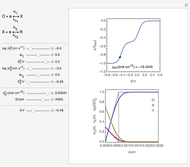
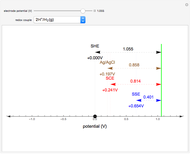
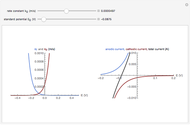
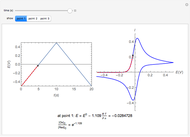
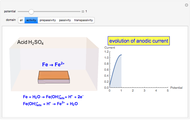
Titrations can be based on redox reactions. The objective here is to do a simple titration and use the titration curve to deduce the concentration and standard potentials of the redox couples—a potentiometric titration. The reaction considered is 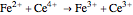 .
.
Contributed by: Quang-Dao Trinh (March 2011)
Open content licensed under CC BY-NC-SA
Snapshots
Details
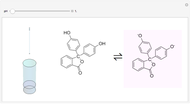
The following shows how to determine equivalence by colorimetry using a color indicator.
With the volume of 
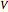 (in mL) added to the solution of
(in mL) added to the solution of 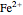 at the unknown initial solution
at the unknown initial solution  , we have the following results:
, we have the following results:

Here 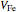 (in mL) is the volume of the solution of
(in mL) is the volume of the solution of 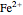 .
.
 (in mol/L) is the unknown concentration of
(in mol/L) is the unknown concentration of 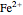 that we need to find.
that we need to find.
 (in mol/L) is the known concentration of
(in mol/L) is the known concentration of  .
.
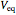 (in mL) is the volume of
(in mL) is the volume of  used to reach equilibrium with
used to reach equilibrium with 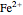 .
.
When  , the concentration of the couple
, the concentration of the couple  is negligible, so the potential measured is that of the couple
is negligible, so the potential measured is that of the couple  /
/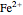 :
:
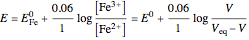 .
.
When  , the couple
, the couple  determines the measured potential:
determines the measured potential:
 .
.
The change of the potential near the equivalence point can be used to determine the equilibrium volume 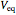 . Then the unknown concentration can be calculated using
. Then the unknown concentration can be calculated using  (mol/L).
(mol/L).
This Demonstration was based on the electrochemistry course (Cours LC102) for first-year students at University Pierre Marie Curie (Paris).
Permanent Citation