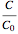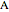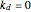Slurry Reactor with Declining Catalyst Activity
Initializing live version

Requires a Wolfram Notebook System
Interact on desktop, mobile and cloud with the free Wolfram Player or other Wolfram Language products.
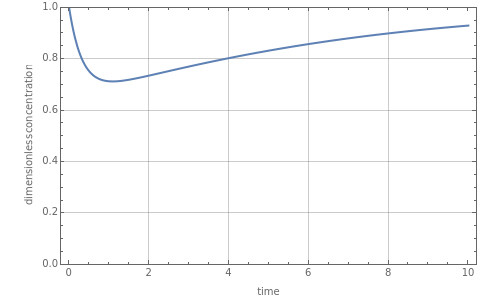
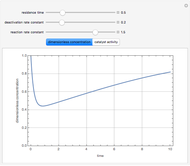
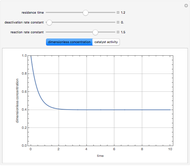
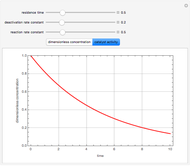
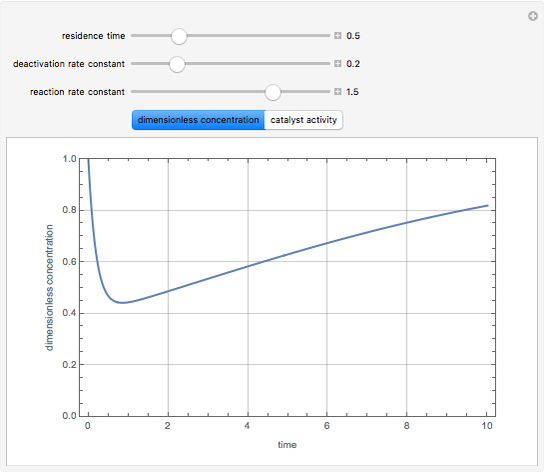
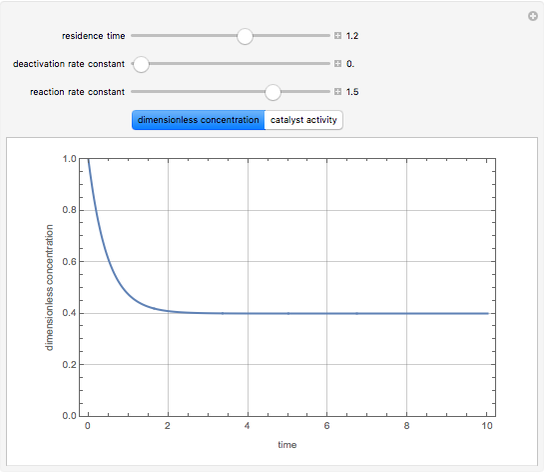
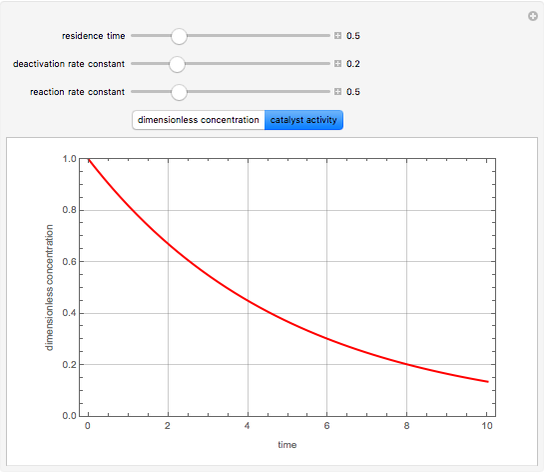
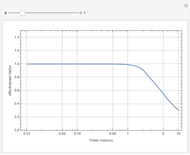
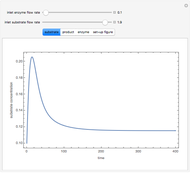
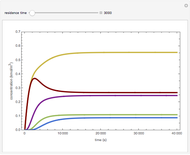
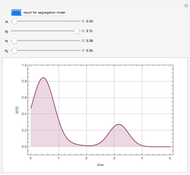
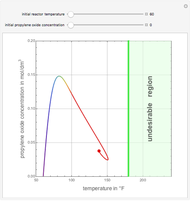
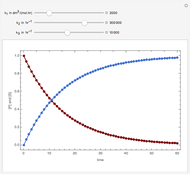
Consider a CSTR (continuous stirred-tank reactor) with a slurried catalyst where a first-order reaction  is taking place. The catalyst is subject to deactivation which follows a first-order law. Thus the catalyst activity,
is taking place. The catalyst is subject to deactivation which follows a first-order law. Thus the catalyst activity,  , obeys the governing equation
, obeys the governing equation 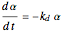 with
with 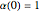 .
.
Contributed by: Housam Binous and Ahmed Bellagi (March 2011)
Open content licensed under CC BY-NC-SA
Snapshots
Details
S. M. Walas, Chemical Reaction Engineering Handbook of Solved Problems, New York: Gordon and Breach Publishers, 1995 p. 727.
Permanent Citation