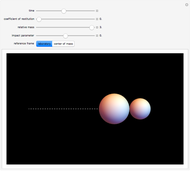
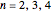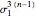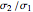Virial Coefficients for a Hard-Sphere Mixture

Requires a Wolfram Notebook System
Interact on desktop, mobile and cloud with the free Wolfram Player or other Wolfram Language products.
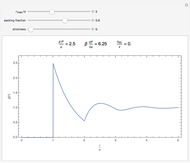
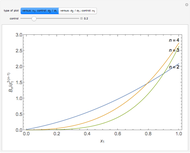
The equation of state of an imperfect gas can be represented as an expansion of the pressure 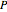 in powers of the density
in powers of the density 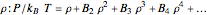 , where
, where 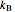 is the Boltzmann constant,
is the Boltzmann constant, 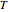 is the absolute temperature, and
is the absolute temperature, and 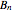 is the
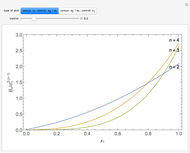
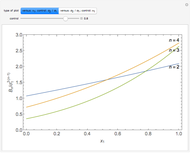
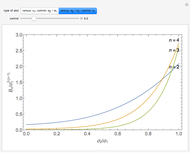
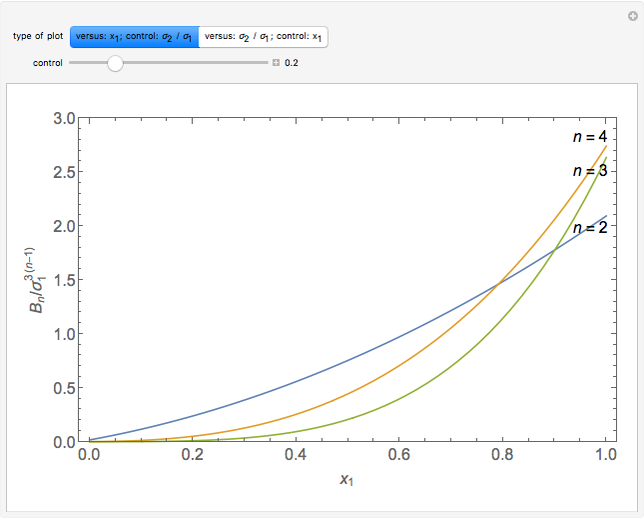
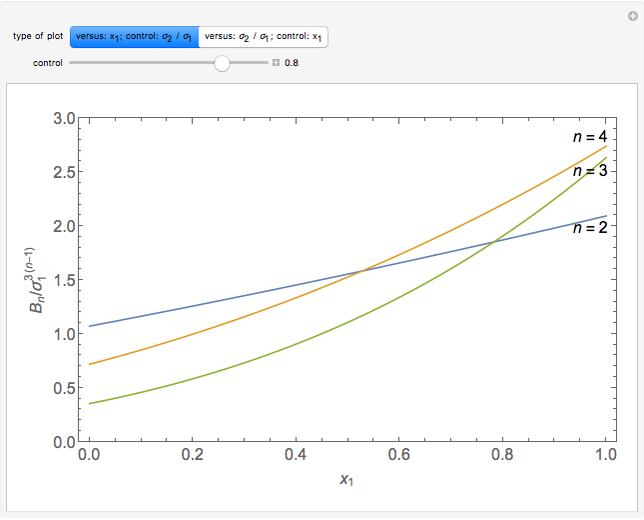
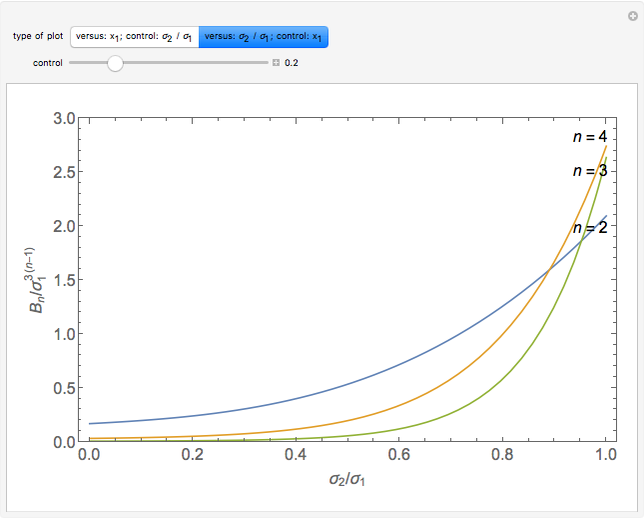
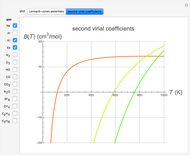
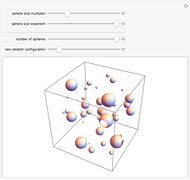
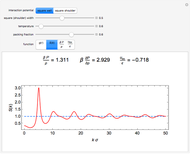
is the 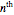 virial coefficient. In the case of a binary mixture of hard spheres, the virial coefficients are functions of the diameters (
virial coefficient. In the case of a binary mixture of hard spheres, the virial coefficients are functions of the diameters (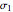 and
and 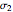 ) of the two components and of the mole fraction
) of the two components and of the mole fraction 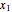 of the larger-sphere component. The coefficients
of the larger-sphere component. The coefficients 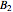 ,
, 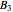 , and all the contributions to
, and all the contributions to 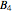 (except one) are known exactly, and an excellent empirical approximattion for the additional contribution to
(except one) are known exactly, and an excellent empirical approximattion for the additional contribution to 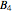 is available.
is available.
Contributed by: Andrés Santos (July 2014)
Open content licensed under CC BY-NC-SA
Snapshots
Details
The analytical expressions for the fourth virial coefficients can be found in [3] and [4].
References
[1] Wikipedia. "Virial Coefficient." (Jun 3, 2014) en.wikipedia.org/wiki/Virial_expansion.
[2] Wikipedia. "Virial Expansion." (Jul 3, 2014) en.wikipedia.org/wiki/Virial_coefficient.
[3] S. Labík and J. Kolafa, "Analytical Expressions for the Fourth Virial Coefficient of a Hard-Sphere Mixture," Physical Review E, 80, 2009 051122. doi:10.1103/PhysRevE.80.051122.
[4] I. Urrutia, "Analytical Behavior of the Fourth and Fifth Virial Coefficients of a Hard-Sphere Mixture," Physical Review E, 84, 2011 062101. doi:10.1103/PhysRevE.84.062101.
Permanent Citation