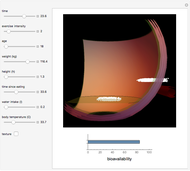
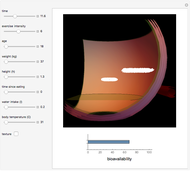
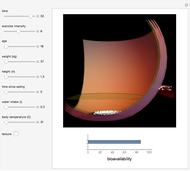
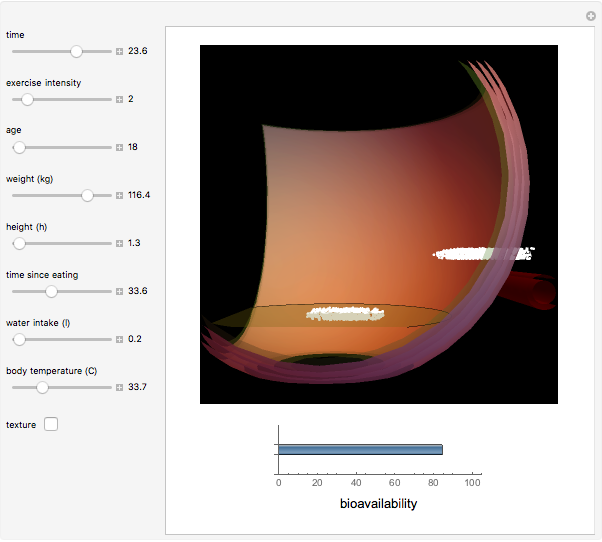
Pharmacokinetic Model for Oral Bioavailability of Aspirin

Requires a Wolfram Notebook System
Interact on desktop, mobile and cloud with the free Wolfram Player or other Wolfram Language products.
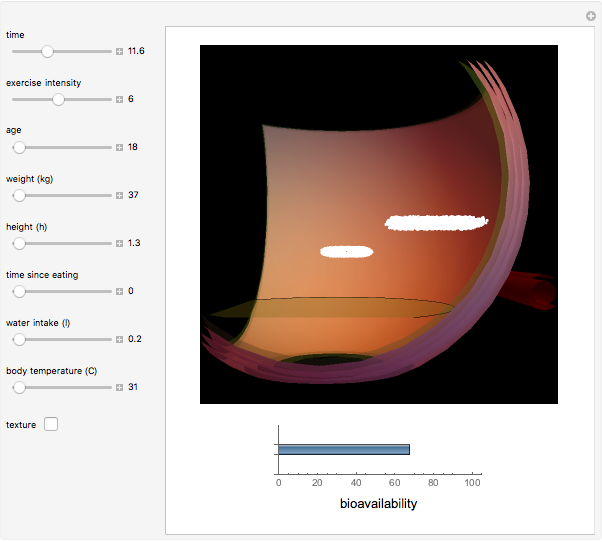
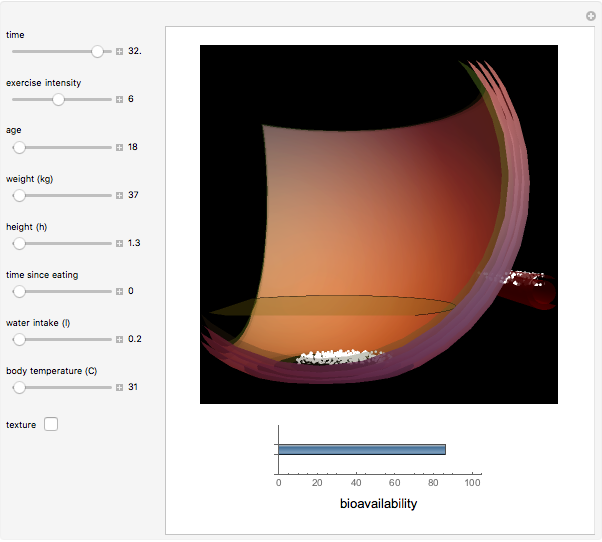
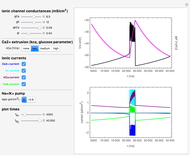
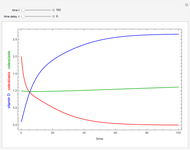
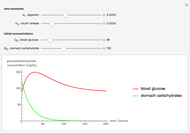
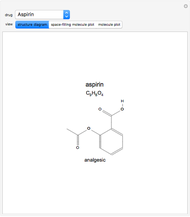
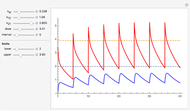
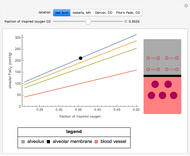
This Demonstration models and simulates the pharmacokinetic process involved in the absorption of aspirin. As aspirin dissolves in the stomach, ionized molecules are secreted while the un-ionized portion of the drug is absorbed into the blood vessel (enlarged for visibility). The bioavailability (the percentage of the drug that is absorbed) is calculated as the drug diffuses across the stomach membrane into the systematic circulation. Using this Demonstration, you can see that factors such as exercise intensity, age, weight, water intake, and so on can substantially affect the bioavailability of the drug, potentially causing the aspirin user to absorb either too little or too much.
Contributed by: Kacey R. Price (September 2014)
(Mathematica Summer Camp 2014)
Open content licensed under CC BY-NC-SA
Snapshots
Details
Pharmacokinetics describe the absorption, distribution, metabolism, and excretion (ADME) process of drugs. To describe the pharmacokinetic process for any particular drug, three major parameters are used: bioavailability, volume of distribution, and clearance.
Physicians prescribe medications to millions of patients to aid in general human health; each patient, however, has their own idiosyncratic traits that can affect how a drug is processed. For example, a person with a heavier body weight will process a drug differently than one with a slimmer build [3]. The effects of age, gender, and weight on pharmacokinetics have been studied thoroughly; effects of activities such as exercise, however, have been neglected thus far. Many athletes take non-steroidal anti-inflammatory drugs (NSAIDs) such as aspirin (acetylsalicylic acid) to relieve soreness. Physical activity is not typically considered when pharmacologists develop a drug, and therefore exercise could cause changes in the ADME process of the drug. Exercise is known to affect a multitude of physiological factors important to pharmacokinetics, including blood distribution, gastrointestinal acidity, gastric emptying, renal excretion rates, plasma protein concentrations, and body heat [3]. Such factors, which are interconnected, affect three primary variables that describe the pharmacokinetics of a drug: bioavailability, volume of distribution (Vd), and clearance [3]. A change in these parameters is known to cause changes in effectiveness of the drug, either causing it to become ineffective, or in contrast, potentially toxic [3]. Unfortunately, current in vivo experimentation of how exercise affects the pharmacokinetics of drugs is very limited. Several types of pharmacokinetic models (including noncompartmental, compartmental, and physiologically based) assist pharmacologists in predicting how the ADME process works in human body when in vivo experimentation on humans is not practical or possible.
Bioavailability, which is the focus point of this Demonstration, describes absorption of the pharmacokinetic process; defined as a percentage, bioavailability is calculated by the amount of drug that reaches the circulation system divided by the amount of drug administered. This parameter can also be calculated by finding the area under the curve (AUC) of plasma concentrations over time; however, for this Demonstration, the former was used. The oral bioavailability of aspirin is considered to be  , meaning the remaining
, meaning the remaining  never reaches the systemic circulation. Bioavailability can be altered, however, by some factors that are affected commonly by physical activity. Exercise can affect the absorption of a drug through increased tissue temperature, blood flow distribution, gastrointestinal acidity, and gastric emptying [5]. As tissue heat increases, molecular movement of tissues will increase, thus causing diffusion to increase across biological membranes [3]. Being absorbed mostly through the gastrointestinal tract, aspirin's absorption is easily affected due to an increase of tissue temperature, which has the potential to increase the bioavailability beyond the intended value [3]. The change in gastric emptying rate can also alter gastric acidity; a change in pH can significantly change drug ionization and thus increase or decrease total absorption [5]. As one exercises, lactic acid is produced that can also decrease the pH of blood and muscle [5]. The gastric mucosa's pH decreases 0.46 pH points during vigorous exercise, thus potentially increasing the absorption of aspirin in the stomach [5]. These potential alterations in absorption ultimately can change the intended bioavailability of a drug such as aspirin. The few studies that have been executed regarding the effect of exercise on drug absorption have yielded conflicting results. In one study of 10 Parkinson's patients, Levodopa was given during an exercise bout; five patients experienced an increase of absorption, three had a decrease of drug absorption, and the remaining two experienced no change. Another study showed that midazolam absorption was slowed significantly during an exercise bout compared to the control non-exercise period. In the same study, however, the absorption of ephedrine was not significantly affected. It was also found that plasma concentration of quinidine, salicylate, and sulfadimidine were not significantly different from the control during a three-hour intermittent exercise bout [3]. Aspirin has not been studied in this respect; due to its common use in athletes, however, it is important that the drug be properly analyzed to observe any possible adverse effects.
never reaches the systemic circulation. Bioavailability can be altered, however, by some factors that are affected commonly by physical activity. Exercise can affect the absorption of a drug through increased tissue temperature, blood flow distribution, gastrointestinal acidity, and gastric emptying [5]. As tissue heat increases, molecular movement of tissues will increase, thus causing diffusion to increase across biological membranes [3]. Being absorbed mostly through the gastrointestinal tract, aspirin's absorption is easily affected due to an increase of tissue temperature, which has the potential to increase the bioavailability beyond the intended value [3]. The change in gastric emptying rate can also alter gastric acidity; a change in pH can significantly change drug ionization and thus increase or decrease total absorption [5]. As one exercises, lactic acid is produced that can also decrease the pH of blood and muscle [5]. The gastric mucosa's pH decreases 0.46 pH points during vigorous exercise, thus potentially increasing the absorption of aspirin in the stomach [5]. These potential alterations in absorption ultimately can change the intended bioavailability of a drug such as aspirin. The few studies that have been executed regarding the effect of exercise on drug absorption have yielded conflicting results. In one study of 10 Parkinson's patients, Levodopa was given during an exercise bout; five patients experienced an increase of absorption, three had a decrease of drug absorption, and the remaining two experienced no change. Another study showed that midazolam absorption was slowed significantly during an exercise bout compared to the control non-exercise period. In the same study, however, the absorption of ephedrine was not significantly affected. It was also found that plasma concentration of quinidine, salicylate, and sulfadimidine were not significantly different from the control during a three-hour intermittent exercise bout [3]. Aspirin has not been studied in this respect; due to its common use in athletes, however, it is important that the drug be properly analyzed to observe any possible adverse effects.
Most models that have been created to predict oral bioavailability of drugs have been based on data obtained from experimentation on animals. Because humans absorb a drug much differently than other species, the results of these models have been relatively inaccurate. For example, a model known as IMPACT-F used this methodology and yielded 27 percentage points of absolute error and a correlation value of  when testing a variety of drugs [2]. After eight years of adjusting the model, the researchers were able to lower the mean absolute error to approximately 15.968 percentage points and a correlation value of
when testing a variety of drugs [2]. After eight years of adjusting the model, the researchers were able to lower the mean absolute error to approximately 15.968 percentage points and a correlation value of  while testing 34 different drugs [2]. In contrast, this Demonstration is focused strictly on physiological changes in the body rather than clinical data to reach a more accurate model.
while testing 34 different drugs [2]. In contrast, this Demonstration is focused strictly on physiological changes in the body rather than clinical data to reach a more accurate model.
References
[1] Bayer Corporation. "Aspirin Comprehensive Prescribing Information." (Jul 30, 2014) www.fda.gov/ohrms/dockets/ac/03/briefing/4012B1_ 03_Appd %201-Professional %20 Labeling.pdf.
[2] D. W. Boomgaarden. "IMPACT-F." Pharmainformatic. (2013) www.pharmainformatic.com/html/prediction_of_f _. html.
[3] T. Buclin, M. Nicod, and S. Kellenberger. "Aspirin Pharmacokinetics." Pharmacokinetics. (Jul 2009) sepia.unil.ch/pharmacology/index.php?id=83.
[4] C. D. Ciccone, "Basic Pharmacokinetics and the Potential Side Effect of Physical Therapy Interventions on Pharmacokinetic Variables," Journal of the American Physical Therapy Association, 75(5), 1995 pp. 343–351.
[5] T. Khazaeinia, A. A. Ramsey, and Y. K. Tam, "The Effects of Exercise on the Pharmokinetics of Drugs," Journal of Pharmacy and Pharmaceutical Sciences, 3(3), 2000 pp. 292–302.
[6] T. L. Lenz, "Pharmacokinetic Drug Interactions with Physical Activity," American Journal of Lifestyle Medicine, 4(3), 2010 pp. 226–229.
Permanent Citation