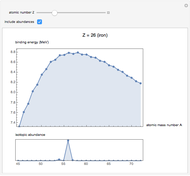
Binding Energies of Isotopes
Initializing live version

Requires a Wolfram Notebook System
Interact on desktop, mobile and cloud with the free Wolfram Player or other Wolfram Language products.
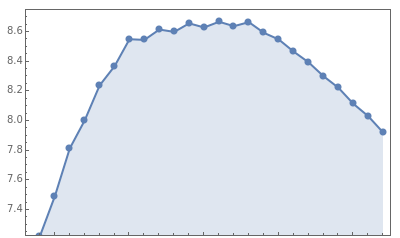
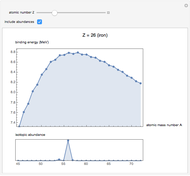
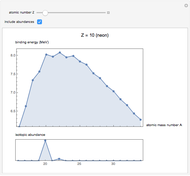
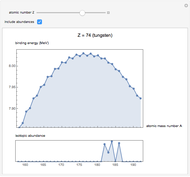
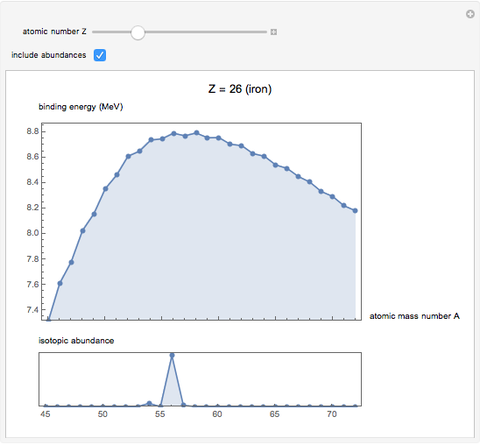
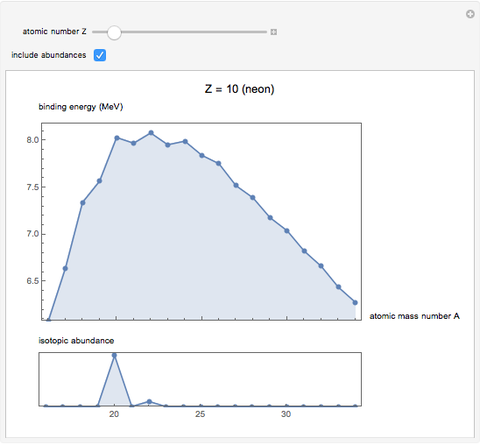
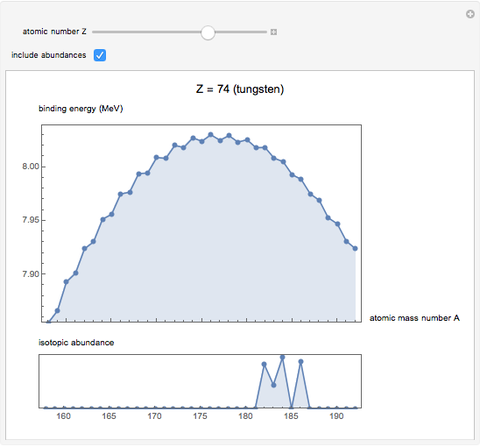
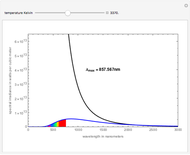
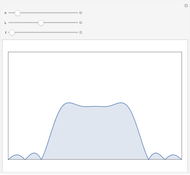
See how binding energies vary with atomic mass number (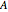 ) for isotopes with different atomic numbers (
) for isotopes with different atomic numbers (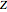 ). The more stable isotopes tend to be the ones with higher binding energies.
). The more stable isotopes tend to be the ones with higher binding energies.
Contributed by: Stephen Wolfram and Jamie Williams (July 2007)
Open content licensed under CC BY-NC-SA
Snapshots
Details
Permanent Citation