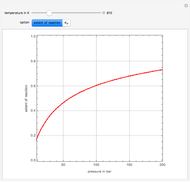
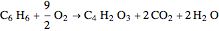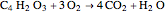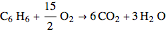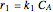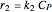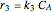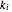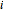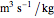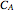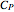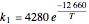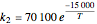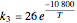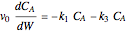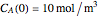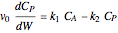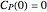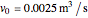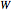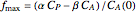Optimizing Maleic Anhydride Production

Requires a Wolfram Notebook System
Interact on desktop, mobile and cloud with the free Wolfram Player or other Wolfram Language products.
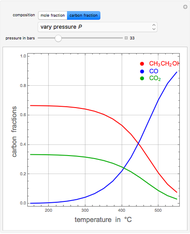
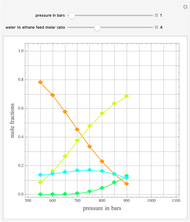
The synthesis of maleic anhydride, 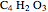 , involves the following reactions in the presence of a vanadium pentoxide catalyst:
, involves the following reactions in the presence of a vanadium pentoxide catalyst:
Contributed by: Housam Binous, Mohammad Mozahar Hossain, and Ahmed Bellagi (December 2015)
Open content licensed under CC BY-NC-SA
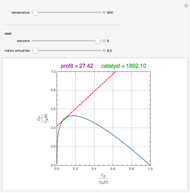
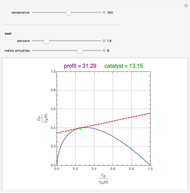
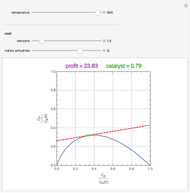
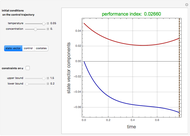
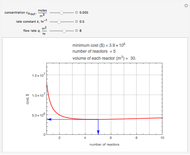
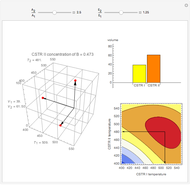
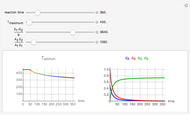
Snapshots
Details
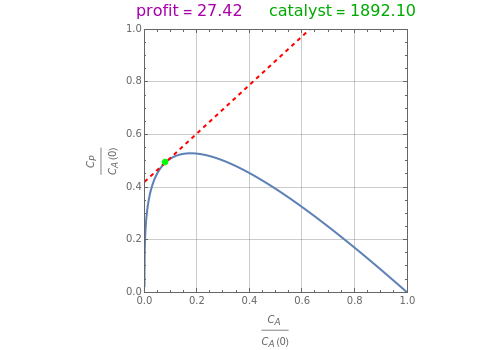
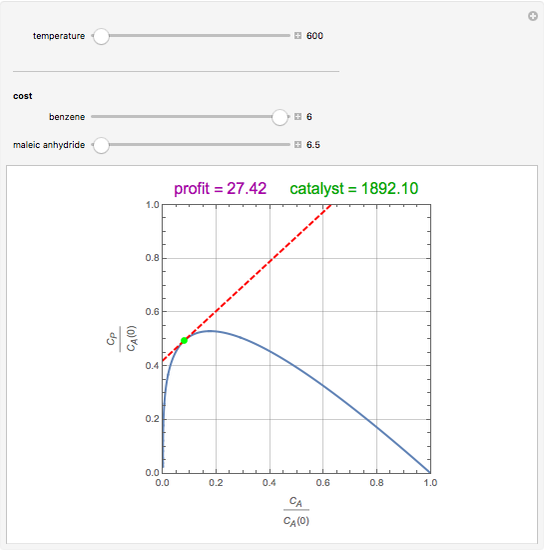
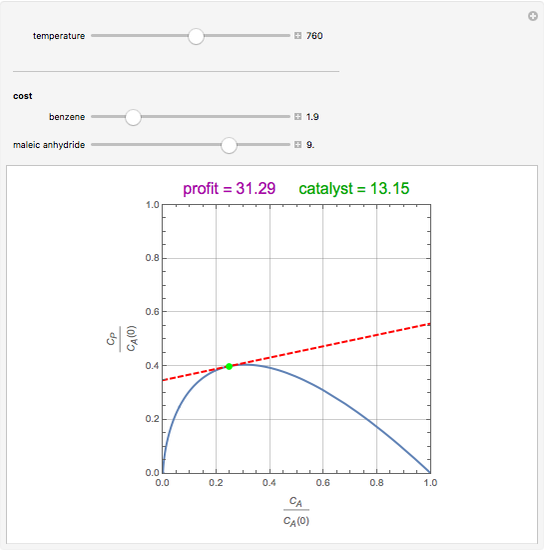
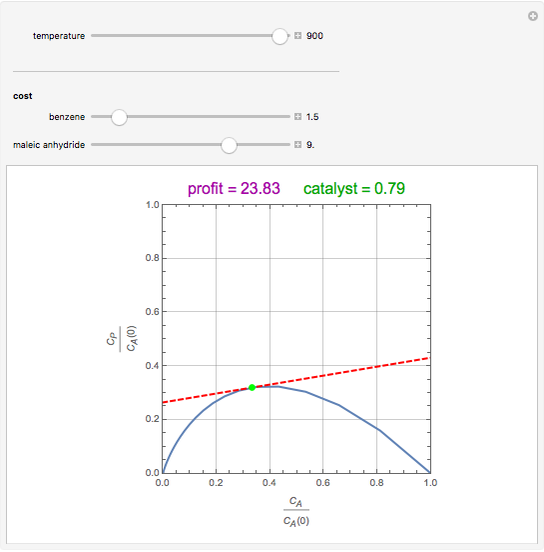
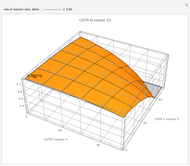
Attainable regions define the achievable compositions that may be obtained from a network of chemical reactions. The attainable region in composition space was first presented by Horn in 1964, with more recent extensions by Glasser and coworkers (1987).
References
[1] F. J. M. Horn, "Attainable Regions in Chemical Reaction Technique," Third European Symposium of Chemical Reaction Engineering, London: Pergamon Press, 1964.
[2] D. Glasser, C. Crowe, and D. Hildebrandt, "A Geometric Approach to Steady Flow Reactors: The Attainable Region and Optimization in Concentration Space," Industrial Engineering & Chemistry Research, 26(9), 1987 pp. 1803–1810.
[3] W. D. Seider, J. D. Seader, and D. R. Lewin, Product and Process Design Principles: Synthesis, Analysis, and Evaluation, 2nd ed., New York: Wiley, 2004.
Permanent Citation