Optimum Temperature for Lactose Hydrolysis in Batch Reactors

Requires a Wolfram Notebook System
Interact on desktop, mobile and cloud with the free Wolfram Player or other Wolfram Language products.
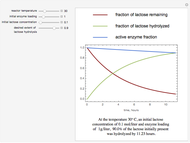
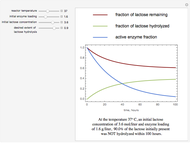
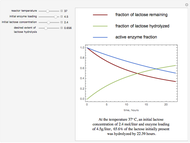
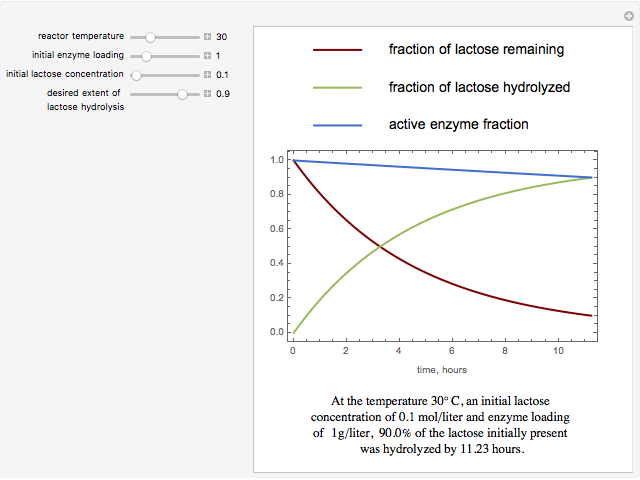
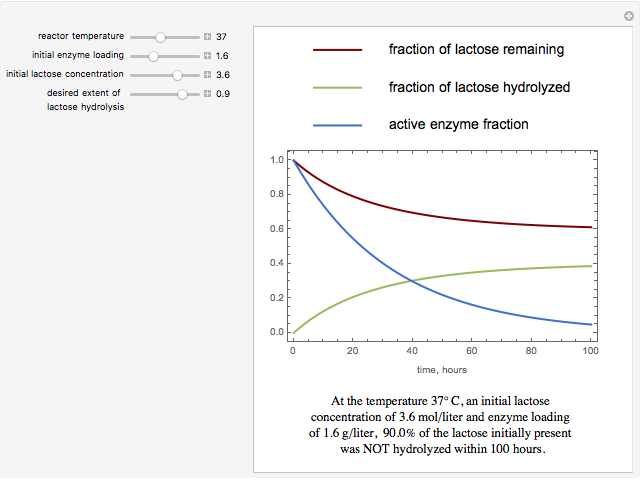
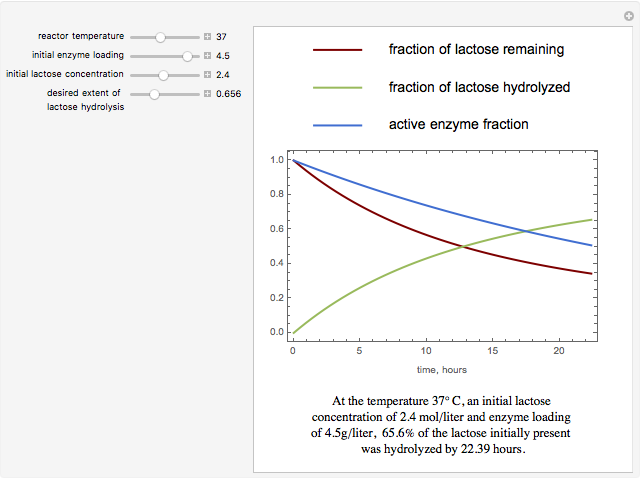
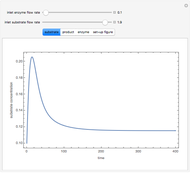
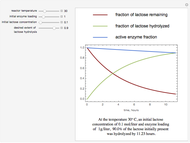
The curves show the time courses of substrate lactose concentration (as a fraction of initial lactose concentration), product galactose concentration (again as a fraction of the initial lactose concentration), and the active fraction of lactase enzyme in a batch reactor for the chosen values of the manipulated variables: reactor temperature (°C), initial enzyme concentration (gram/liter), and initial lactose concentration (mol/liter).
[more]
Contributed by: David J. W. Simpson and Dhinakar S. Kompala (March 2011)
Open content licensed under CC BY-NC-SA
Snapshots
Details
Lactose present in milk or some other dairy products like ice cream is not digested by a significant fraction of the human population (3 to 70% depending on the populational group), creating various lactose intolerance symptoms, including diarrhea, bloating, flatulence, and so on. An enzyme known as lactase or beta-galactosidase can hydrolyze the milk sugar lactose into two more easily digested sugars, glucose and galactose. One of the two product sugars, galactose, is usually a competitive inhibitor of the lactase enzyme.
At higher temperatures, the rate of lactose hydrolysis is faster initially, but the enzyme gets degraded faster. If the enzyme is completely degraded within a short time due to higher reactor temperatures, the desired level of hydrolysis (usually 99% or above) may not be achieved with the initial loading of enzyme. At lower temperatures, more of the enzyme remains active for a longer period, but the rate of lactose hydrolysis is slower. Therefore, it takes longer to achieve the desired level of lactose hydrolysis. At some intermediate temperature, we might find the time to achieve the desired level of lactose hydrolysis is minimized for a fixed amount of the initial enzyme loading.
In simple batch reactors, the initial enzyme concentration or loading can be increased to achieve faster or more complete hydrolysis, but the enzyme is expensive. Therefore, we may want to achieve the desired hydrolysis level with the minimum amount of enzyme loading possible. A practical objective function may include minimizing a cost-weighted combination of enzyme loading and reaction time.
The kinetic equations used in the above simulation are taken from: E. Jurado, F. Camacho, G. Luzon, and J. M. Vicaria, "A New Kinetic Model Proposed for Enzymatic Hydrolysis of Lactose by a Beta-Galactosidase from Kluyveromyces fragilis," Enzyme and Microbial Technology 31(3), 2002 pp. 300–309.
Permanent Citation
"Optimum Temperature for Lactose Hydrolysis in Batch Reactors"
http://demonstrations.wolfram.com/OptimumTemperatureForLactoseHydrolysisInBatchReactors/
Wolfram Demonstrations Project
Published: March 7 2011