Surface Tension: Walking on Water

Requires a Wolfram Notebook System
Interact on desktop, mobile and cloud with the free Wolfram Player or other Wolfram Language products.
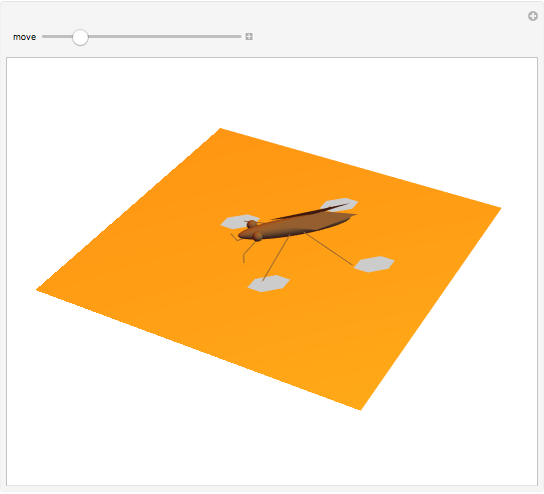
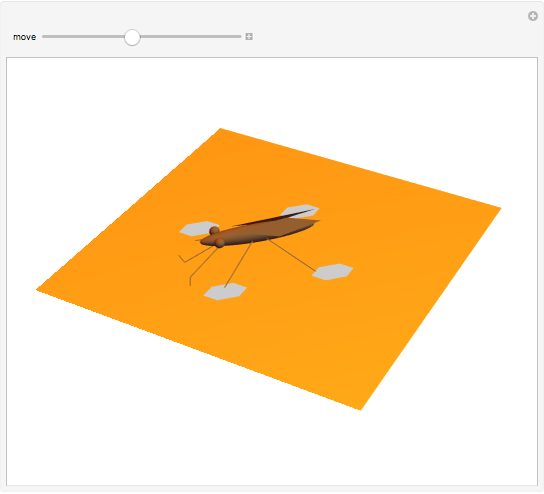
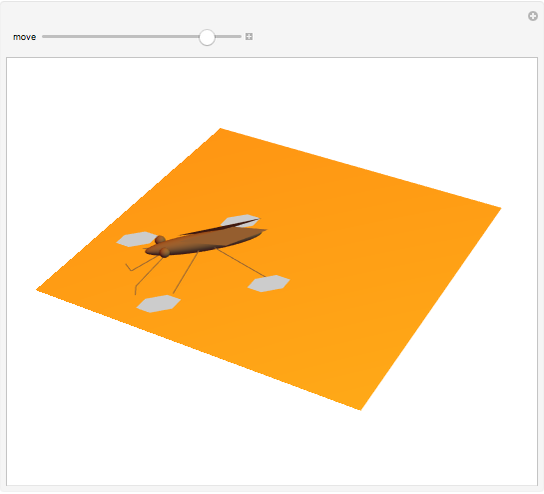
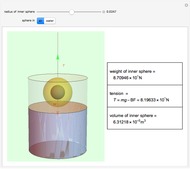
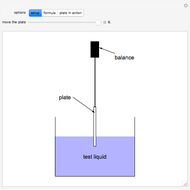
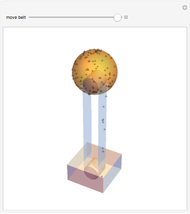
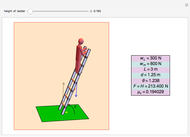
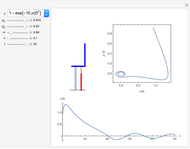
The cohesive forces between the molecules of a liquid force the surface to contract to the smallest possible area. This makes the surface of the liquid behave like a membrane, so that light objects like leaves or insects can float. Surface tension explains the almost perfectly spherical form of falling drops.
Contributed by: Enrique Zeleny (March 2011)
Open content licensed under CC BY-NC-SA
Snapshots
Details
detailSectionParagraphPermanent Citation
"Surface Tension: Walking on Water"
http://demonstrations.wolfram.com/SurfaceTensionWalkingOnWater/
Wolfram Demonstrations Project
Published: March 7 2011