Effects of Cell and Solution Concentrations on Osmosis

Requires a Wolfram Notebook System
Interact on desktop, mobile and cloud with the free Wolfram Player or other Wolfram Language products.
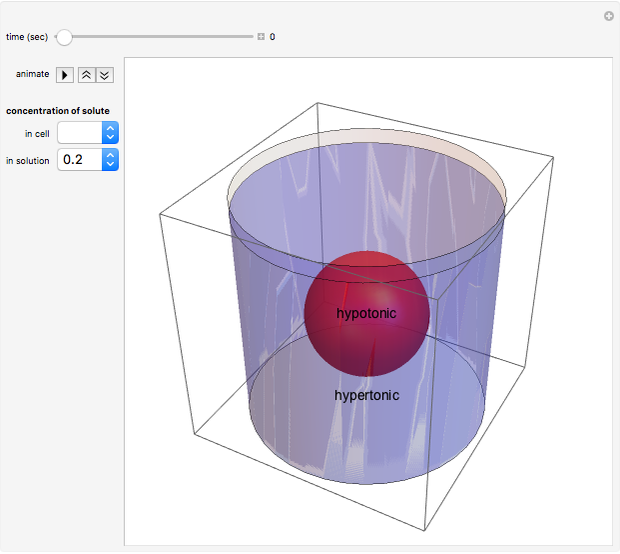
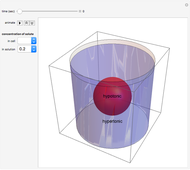
This Demonstration illustrates the biological concept of tonicity, a measure of the osmotic pressure gradient between a cell and its surrounding solution. Water can travel across a cell membrane through a process called osmosis, and it always moves down its concentration gradient. Therefore, water will travel from a hypotonic solution, which has high water concentration (and low solute concentration) to a hypertonic solution, which has low water concentration (and high solute concentration). This Demonstration shows the movement of water for different tonicities.
Contributed by: Aileen Jong and Lizzie Neal (June 2013)
With additional contributions by: Ben Lewis
Open content licensed under CC BY-NC-SA
Snapshots
Details
A solute is a substance dissolved in a liquid, the solvent: water in this Demonstration.
Osmosis is the movement of water across a cell membrane. Cells use osmosis to maintain concentration equilibrium (the concentrations of solute inside and outside the cell are equal). Changing the amount of water allows the cells to achieve equilibrium. When a cell is placed in a solution in which the concentrations are not the same as in the cell, the cell undergoes osmosis. The water travels down the concentration gradient from higher water concentration (and lower solute concentration) to lower water concentration (and higher solute concentration). In other words, water moves from a hypotonic region to a hypertonic region. Determining which fluid is hypotonic and hypertonic is relative.
When water leaves a cell, it shrinks, which is called plasmolysis. When water enters a cell, it expands, which creates turgor pressure on the walls of a plant cell and can cause the cell to explode.
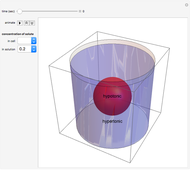
In these snapshots, the solute concentration in the solution exceeds the solute concentration in the cell.
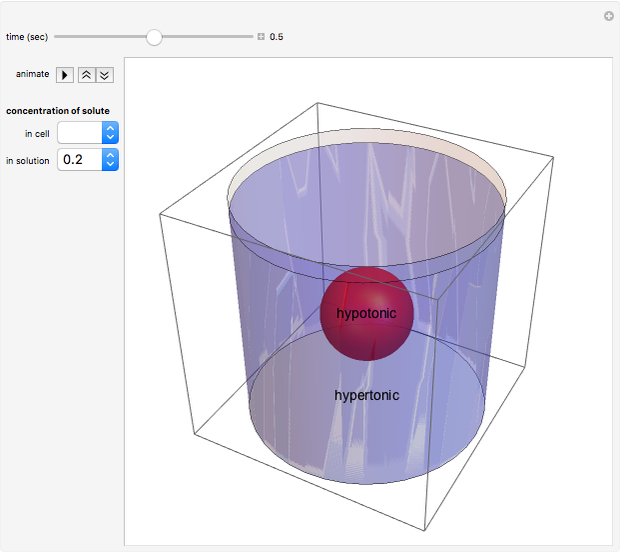
Snapshot 1: a hypotonic cell (smaller solute concentration, more water) is initially placed in a hypertonic solution (greater solute concentration, less water)
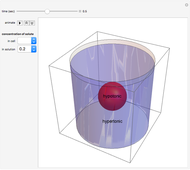
Snapshot 2: at 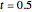 water travels across the cell membrane from the hypotonic to hypertonic location, implying that water leaves the cell, causing it to shrink
water travels across the cell membrane from the hypotonic to hypertonic location, implying that water leaves the cell, causing it to shrink
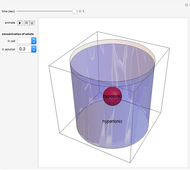
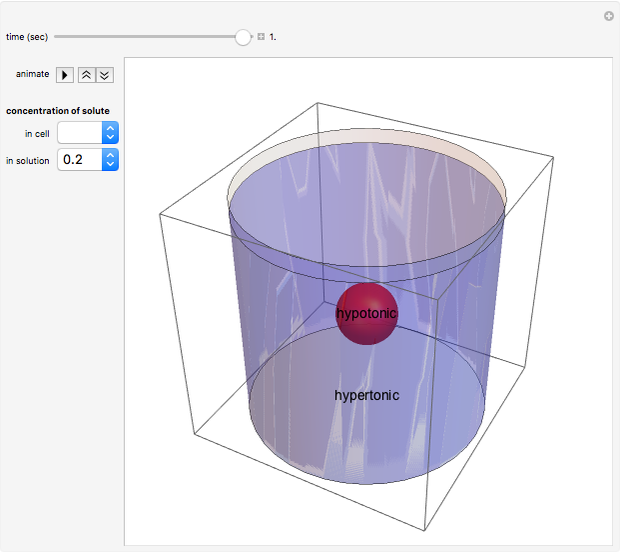
Snapshot 3: the cell continues to shrink (undergo plasmolysis) until it reaches equilibrium
The opposite would occur if the solute concentration in the cell exceeded the solute concentration in the solution.
Special thanks to the University of Illinois NetMath Program and the mathematics department at William Fremd High School.
References
[1] N. A. Campbell et al., AP Edition Biology, 8th ed., New York: Pearson/Benjamin Cummings, 2008.
[2] Hartnell College. "Hartnell College Biology Tutorials." (May 25, 2013) www.hartnell.edu/biology-tutorials.
Permanent Citation
"Effects of Cell and Solution Concentrations on Osmosis"
http://demonstrations.wolfram.com/EffectsOfCellAndSolutionConcentrationsOnOsmosis/
Wolfram Demonstrations Project
Published: June 3 2013