Adsorption Isotherms of Ethylene in NaX (Zeolite Structure FAU)
Initializing live version

Requires a Wolfram Notebook System
Interact on desktop, mobile and cloud with the free Wolfram Player or other Wolfram Language products.
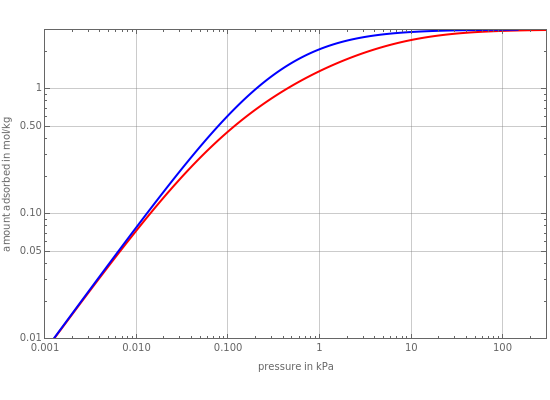
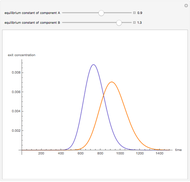
This Demonstration plots adsorbtion versus pressure,  , at various temperatures for ethylene (
, at various temperatures for ethylene ( ) in NaX, known as faujasite zeolite structure (FAU). The amount adsorbed,
) in NaX, known as faujasite zeolite structure (FAU). The amount adsorbed,  (expressed in mol/kg), increases at low temperatures and high pressures. The adsorption isotherm, shown in red, is given by the following relation:
(expressed in mol/kg), increases at low temperatures and high pressures. The adsorption isotherm, shown in red, is given by the following relation:
Contributed by: Housam Binous (March 2011)
Open content licensed under CC BY-NC-SA
Snapshots
Details
T. M. Letcher, ed., Chemical Thermodynamics for Industry, Cambridge: The Royal Society of Chemistry, 2004.
Permanent Citation