Chemical Reaction Trajectories Using Bézier Curves

Requires a Wolfram Notebook System
Interact on desktop, mobile and cloud with the free Wolfram Player or other Wolfram Language products.
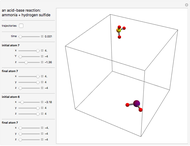
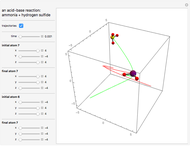
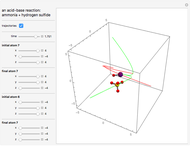
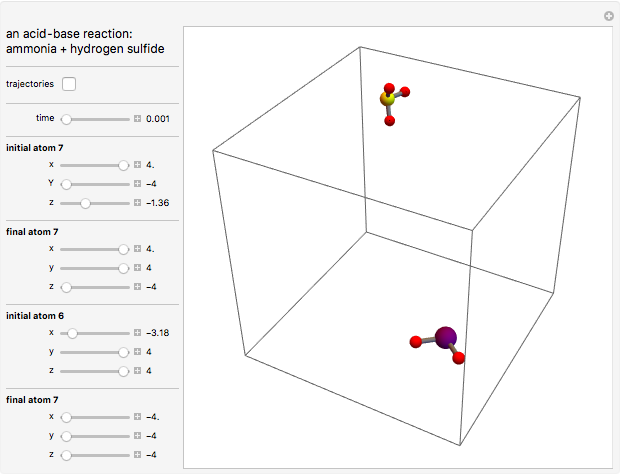
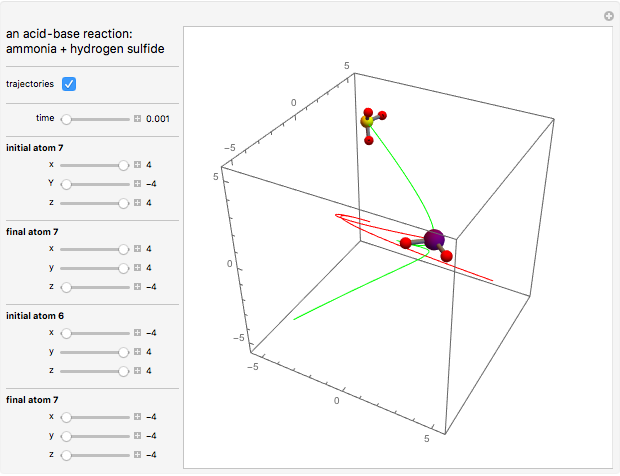
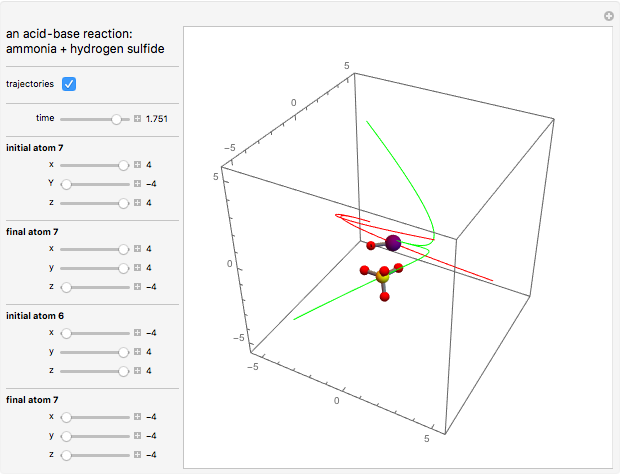
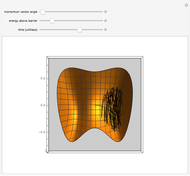
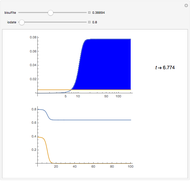
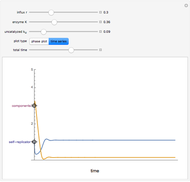
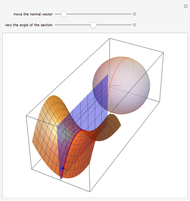
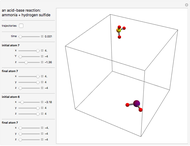
This Demonstration shows an example of a simple chemical reaction, which might include a proton transfer or an acid-base reaction. Specifically, we consider the reaction between ammonia (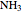 ) and hydrogen sulfide (
) and hydrogen sulfide (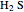 ) to produce an ammonium ion (
) to produce an ammonium ion (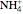 ) and bisulfide ion (
) and bisulfide ion (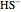 ).
).
Contributed by: Mark Mochalsky, Pascual Lahuerta, Jose Vicente Beltran, and Juan Monterde (September 2012)
(University of Valencia – Spain)
Open content licensed under CC BY-NC-SA
Snapshots
Details
detailSectionParagraphPermanent Citation
"Chemical Reaction Trajectories Using Bézier Curves"
http://demonstrations.wolfram.com/ChemicalReactionTrajectoriesUsingBezierCurves/
Wolfram Demonstrations Project
Published: September 13 2012