Embryonic Cleavage

Requires a Wolfram Notebook System
Interact on desktop, mobile and cloud with the free Wolfram Player or other Wolfram Language products.
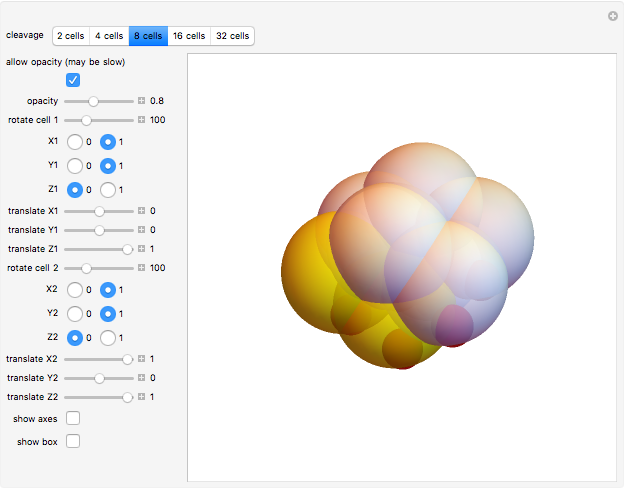
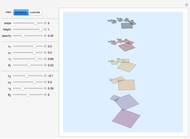
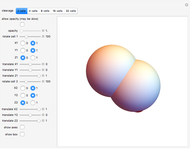
This Demonstration shows an implementation of a three-dimensional substitution system to model the early phase of embryonic cleavage. Play with the substitution rule, and discover how many kind of embryos can be described in a simple way!
Contributed by: Luca Zammataro (March 2011)
Open content licensed under CC BY-NC-SA
Snapshots
Details
Embryonic cleavage depends on the distribution in the cytosol of molecular signals that influence mitotic spindle positioning. This Demonstration proposes an implementation of a three-dimensional substitution system computational model (3DSS) to model the early phase of embryonic cleavage. Substitution systems are simple programs in which at each step every element is replaced by a new block of elements, independently of the state of any neighboring element.
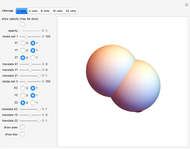
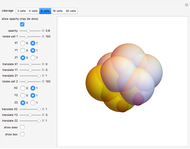
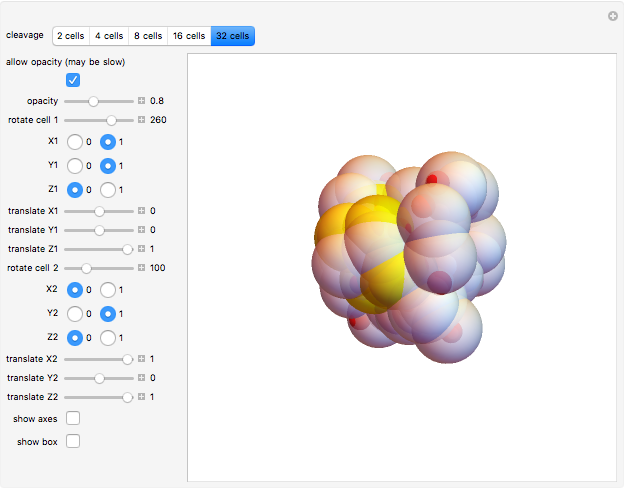
Default parameters refer to mechanisms of early embryonic cleavage dynamics in the ascidian Styela Partita. The model shows that the same spindle pole orientation rule governs the first five embryonic cleavages, which progressively allow the transition from one to 32 daughter cells.
This demonstrates how a complex pattern, the 32-cells embryo, can be obtained by repeatedly applying a simple geometrical rule. The 3DSS algorithm shown here (which is a three-dimensional implementation of classic substitution systems) could prove useful for shedding light on the dynamics underlying embryonic patterning (see the snapshots). Indeed, embryonic cleavage could be explained as an iterated process of a certain rule generated by the asymmetric distribution of transmembrane and cytoplasmic proteins that anchor and orient the spindle at each division cycle. In this simulation, a sphere, representing the fertilized egg, undergoes progressive segmentation. The first cell is cleaved into two distinct daughter cells, according to a simple rule that reproduces the coordinates of their spindle poles and the angle of the mitotic spindle by geometrical displacement of the second daughter cell with respect to the first one.
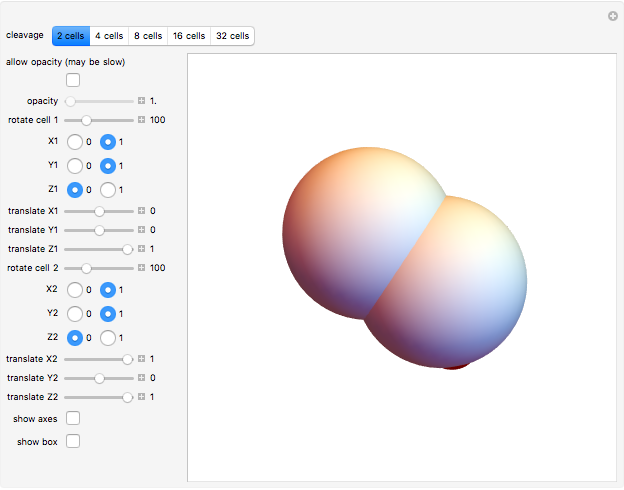
The Mathematica functions used in this Demonstration are Rotate and Translate. Change the roto-translation rule for each couple of daughter cells, moving the rotation degree control bars and the translate bars for the  ,
,  , and
, and  axes.
axes.
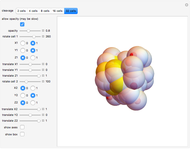
A crucial issue during embryonic development is represented by cell fate and differentiation. In the Styela Partita embryo, the tadpole tail muscle cells originate from a pair of lower posterior cells, which appear at the eight-cell stage (third cleavage). Cells descending from these muscle cell progenitors are easily identified since they contain a yellow cytoplasmic pigment, which is enriched in the zinc finger transcription factor Macho-1 that triggers a regulatory cascade driving localized expression of a group of genes to initiate muscle differentiation.
In this Demonstration, the tadpole tail muscle cells are in yellow; red spheres inside cells can help you in visualizing the roto-translation tracking when you modify the rule.
The fact that this 3DSS fully recapitulates the dynamics of muscle cell progeny indicates that the reiteration of a single rule controlling spindle axis orientation is responsible for embryonic development not only in terms of shape, but also in terms of cell differentiation.
References:
L. Zammataro, G. Serini, T. Rowland, and F. Bussolino, "Embryonic Cleavage Modeling as a Computational Approach to Sphere Packing Problem," Journal of Theoretical Biology, 245(1), 2006 pp. 77–82.
Permanent Citation
"Embryonic Cleavage"
http://demonstrations.wolfram.com/EmbryonicCleavage/
Wolfram Demonstrations Project
Published: March 7 2011