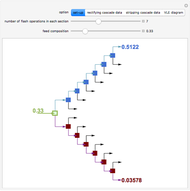
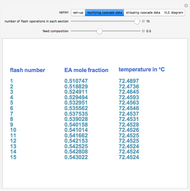
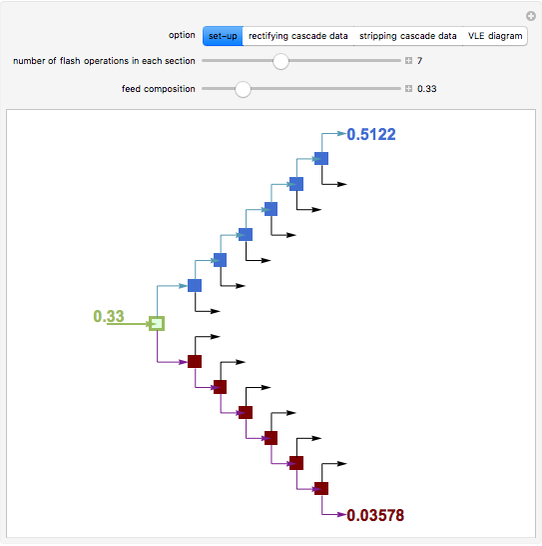
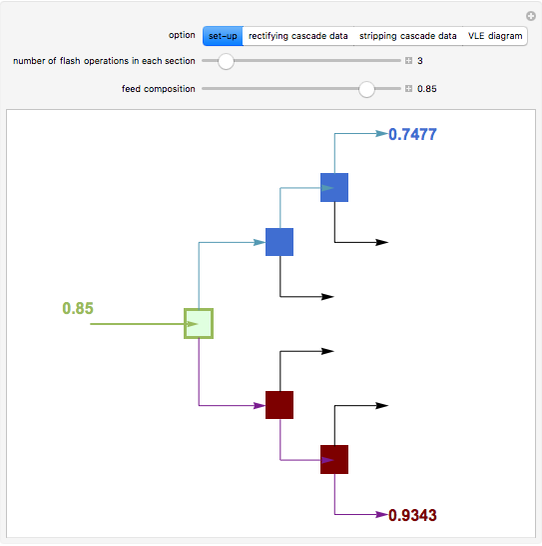
Flash Distillation Cascade for an Ethyl Acetate-Ethanol Mixture
Initializing live version

Requires a Wolfram Notebook System
Interact on desktop, mobile and cloud with the free Wolfram Player or other Wolfram Language products.
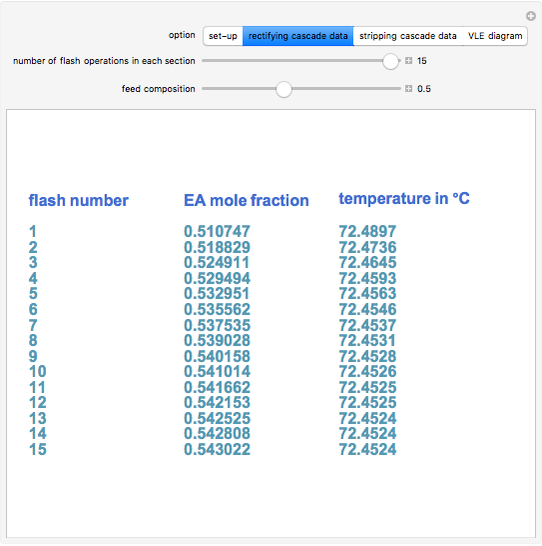
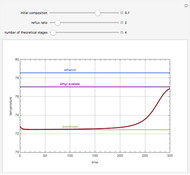
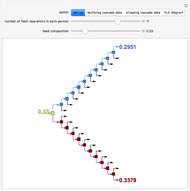
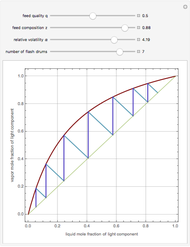
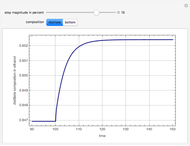
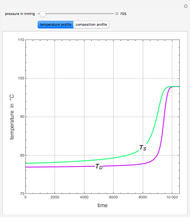
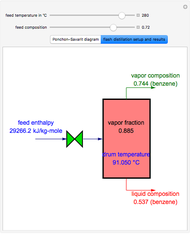
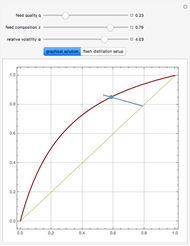
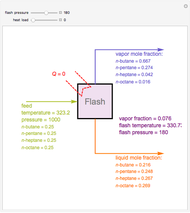
A binary mixture, composed of ethyl acetate and ethanol, is fed to a flash distillation cascade. The mixture presents a positive azeotrope with 0.54 mole fraction ethyl acetate at a boiling point of 72.4°C. Vapor-liquid equilibrium data for the simulations are generated using a modified Raoult's law with activity coefficients predicted by the van Laar model. The user specifies the feed mixture composition as mole % ethyl acetate (labeled EA in the data tables).
[more]
Contributed by: Housam Binous and Ahmed Bellagi (March 2011)
Open content licensed under CC BY-NC-SA