Minimized Volume for Reactors in Series

Requires a Wolfram Notebook System
Interact on desktop, mobile and cloud with the free Wolfram Player or other Wolfram Language products.
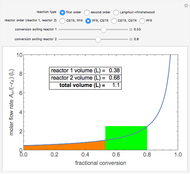
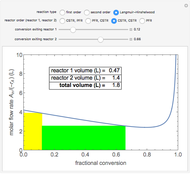
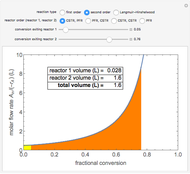
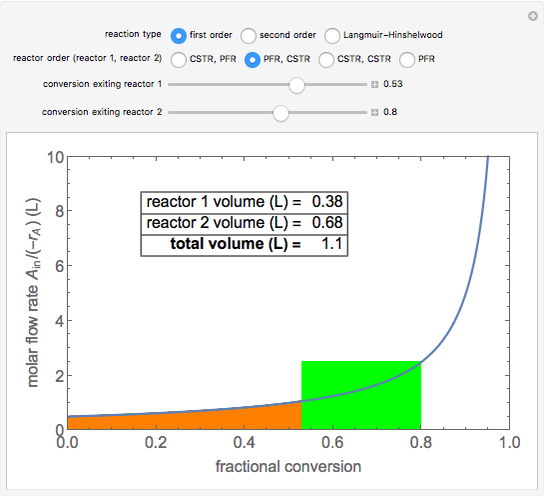
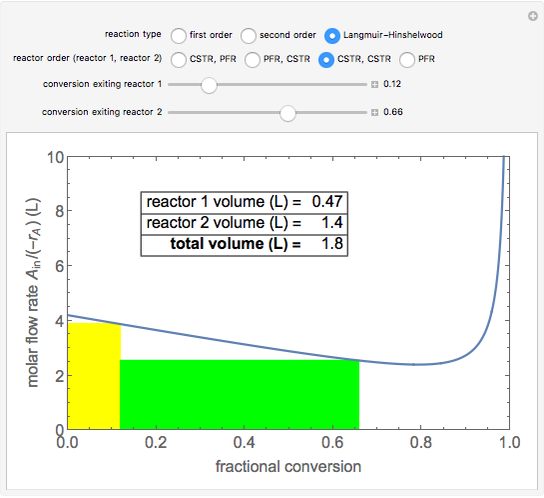
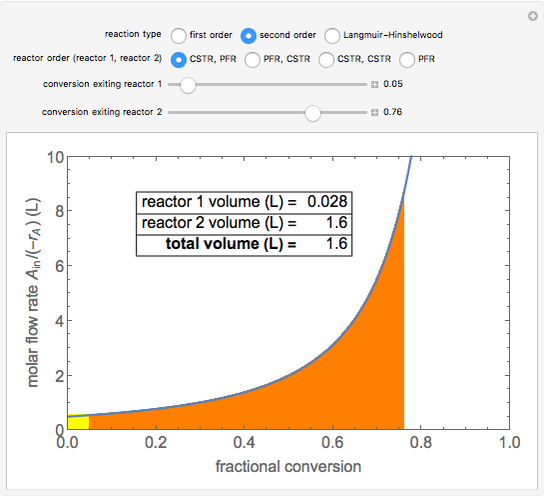
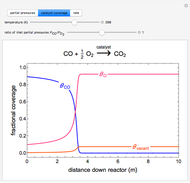
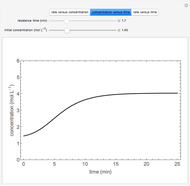
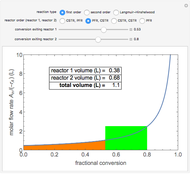
A plot of (feed molar flow rate)/(rate of reaction) versus fractional conversion is used to demonstrate how to minimize total reactor volume for two isothermal reactors in series. Select the order of the plug flow reactor (PFR) and continuous stirred-tank reactor (CSTR) with buttons. Use buttons to select the reaction kinetics for  ; the reaction can be first order, second order or a Langmuir–Hinshelwood mechanism
; the reaction can be first order, second order or a Langmuir–Hinshelwood mechanism  . The colored areas are proportional to the reactor volumes. The PFR volume is proportional to the area under the curve (from inlet to outlet conversion), and the CSTR volume is the difference between inlet and outlet conversion multiplied by the
. The colored areas are proportional to the reactor volumes. The PFR volume is proportional to the area under the curve (from inlet to outlet conversion), and the CSTR volume is the difference between inlet and outlet conversion multiplied by the  axis ratio at the reactor outlet. Use sliders to select the fractional conversion exiting each reactor.
axis ratio at the reactor outlet. Use sliders to select the fractional conversion exiting each reactor.
Contributed by: Nicholas R. Larson (May 2014)
With additional contributions by: Rachael L. Baumann, John L. Falconer and Nick Bongiardina
(University of Colorado Boulder, Department of Chemical and Biological Engineering)
Open content licensed under CC BY-NC-SA
Snapshots
Details
The reactor volumes for a continuous stirred-tank reactor (CSTR) and plug flow reactor (PFR) are:
 ,
,
 ,
,
where  is the inlet molar flow rate of reactant
is the inlet molar flow rate of reactant  ,
,  is the rate of disappearance of reactant
is the rate of disappearance of reactant  and
and  and
and  are the inlet and outlet reactor conversions.
are the inlet and outlet reactor conversions.
Assuming constant reactor inlet flow rate 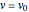 , the molar flow rate of
, the molar flow rate of  is
is  . The concentration of
. The concentration of  can be written in terms of conversion
can be written in terms of conversion  .
.
The reaction rates for the different reaction types are:
first order:  ,
,
second order:  ,
,
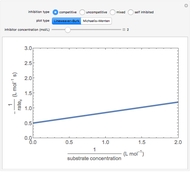
Langmuir–Hinshelwood:  ,
,
assuming  , the Langmuir–Hinshelwood rate law can be written in terms of conversion
, the Langmuir–Hinshelwood rate law can be written in terms of conversion  :
:
 ,
,
where  is the rate constant, and
is the rate constant, and  and
and  are the adsorption equilibrium constants for
are the adsorption equilibrium constants for  and
and  .
.
The screencast video at [2] explains how to use this Demonstration.
References
[1] H. S. Fogler, Essentials of Chemical Reaction Engineering, 1st ed., Upper Saddle River, NJ: Pearson Education, 2010 pp. 33–82, 445.
[2] Minimized Volume for Reactors in Series [Video]. (Sep 12, 2016) www.colorado.edu/learncheme/kinetics/MinVolumeForReactorsInSeries.html.
Permanent Citation