Methyl Tert-Butyl Ether (MTBE) Synthesis with a Reactive Distillation Unit
Initializing live version

Requires a Wolfram Notebook System
Interact on desktop, mobile and cloud with the free Wolfram Player or other Wolfram Language products.
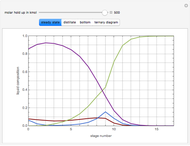
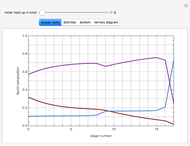
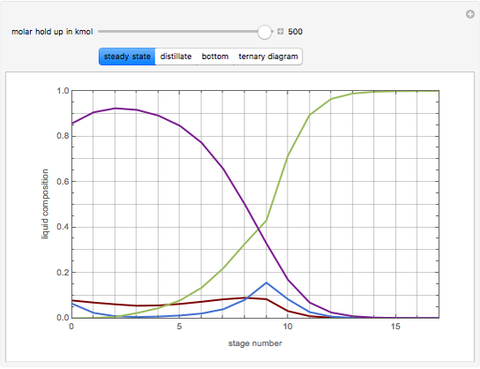
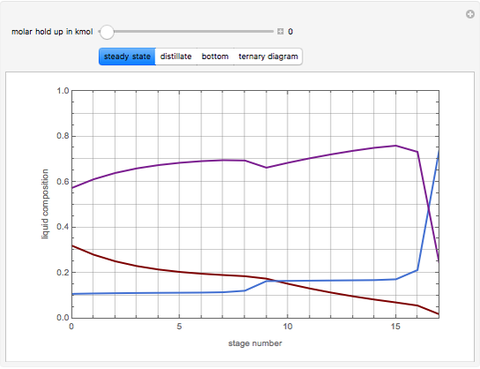
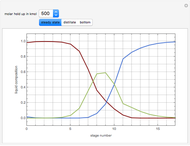
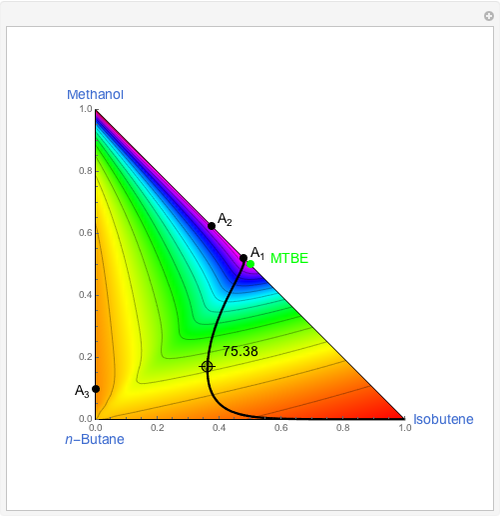
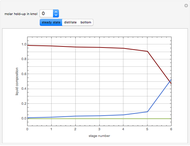
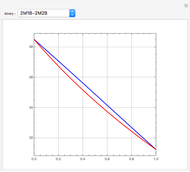
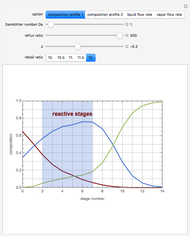
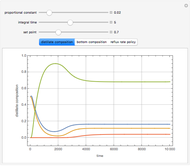
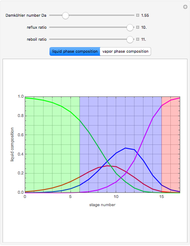
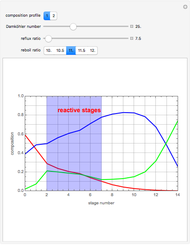
This Demonstration simulates a reactive distillation column that produces methyl tert-butyl ether (MTBE).
[more]
Contributed by: Housam Binous, Manel Selmi, Ines Wada, Selima Allouche and Ahmed Bellagi (March 2011)
Open content licensed under CC BY-NC-SA
Snapshots
Details
F. Chen, R. S. Huss, M. F. Malone, and M. F. Doherty, "Simulation of Kinetic Effects in Reactive Distillation," Computers and Chemical Engineering, 24(11), 2000 pp. 2457–2472.
Permanent Citation