Liquid-Liquid Miscibility for Organic Acids with Methanol

Requires a Wolfram Notebook System
Interact on desktop, mobile and cloud with the free Wolfram Player or other Wolfram Language products.
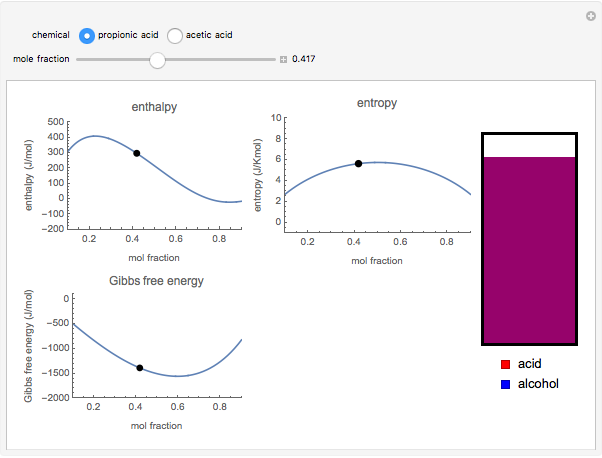
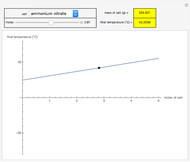
Liquid miscibility is the measure of how well a pair of liquids mixes/dissolves. The miscibility of two liquids depends on the Gibbs free energy of the system, which is a function of temperature, enthalpy 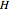 and entropy
and entropy 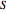 . The difference in free energy between the mixture and its pure components is given by
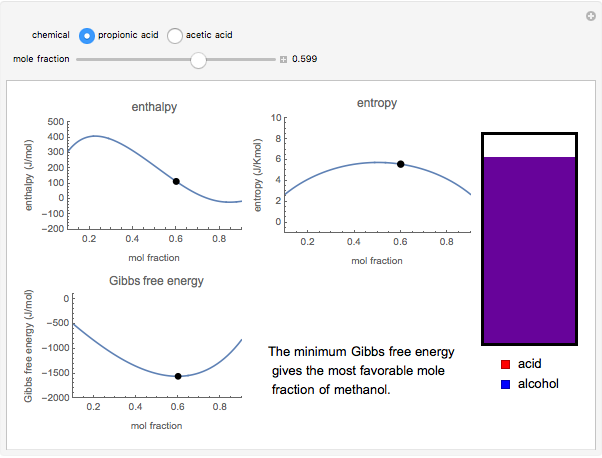
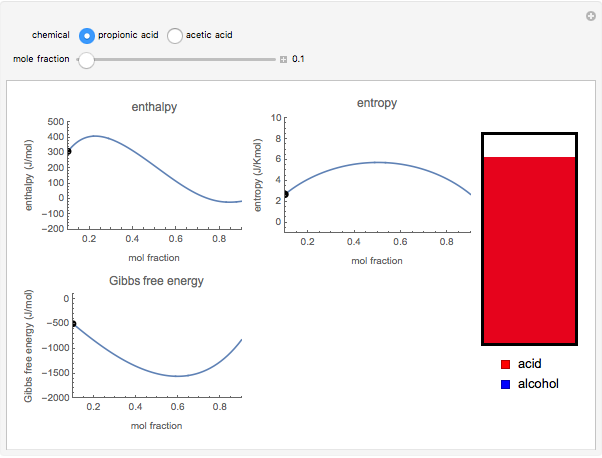
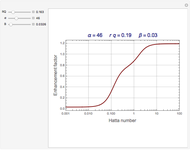
. The difference in free energy between the mixture and its pure components is given by 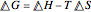 . In the two-component systems modeled in this Demonstration, increasing or decreasing the mole fraction of one component such that the total number of moles always adds up to 1 changes the enthalpy and entropy of the system. This in turn changes the free energy of the system. This Demonstration assumes constant temperature, and shows two examples: methanol/propionic acid and methanol/acetic acid. When the mole fraction of methanol, which can be manipulated with the slider, gives the lowest
. In the two-component systems modeled in this Demonstration, increasing or decreasing the mole fraction of one component such that the total number of moles always adds up to 1 changes the enthalpy and entropy of the system. This in turn changes the free energy of the system. This Demonstration assumes constant temperature, and shows two examples: methanol/propionic acid and methanol/acetic acid. When the mole fraction of methanol, which can be manipulated with the slider, gives the lowest 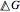 , the system is thermodynamically in the most stable state.
, the system is thermodynamically in the most stable state.
Contributed by: Ruchita Iyer and Soundharya Subramaniam (December 2017)
Additional contributions by: Eitan Geva (University of Michigan)
Open content licensed under CC BY-NC-SA
Details
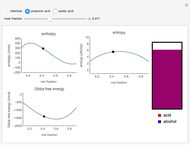
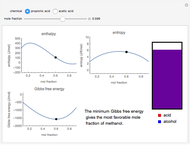
Snapshot 1: the most favorable state of propionic acid and methanol as Gibbs free energy is minimized
Snapshot 2: the mole fraction of methanol is equal to .1; this can be described as a solution of methanol in propionic acid
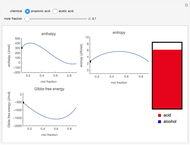
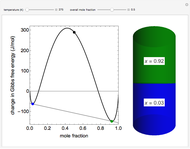
Snapshot 3: the mole fraction of methanol is equal to .9, and is therefore a solution of propionic acid in methanol
Reference
[1] R. Haase and R. Lorenz, "Enthalpies of Mixing for Binary Liquid Mixtures of Monocarbonic Acids and Alcohols," Zeitschrift für Naturforschung A, 40(9), 1985 pp. 947–951.
Submission from the Compute-to-Learn course at the University of Michigan.
Snapshots
Permanent Citation