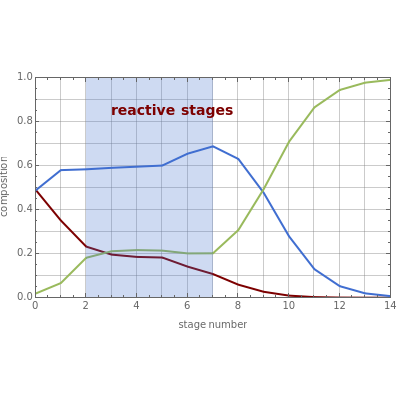
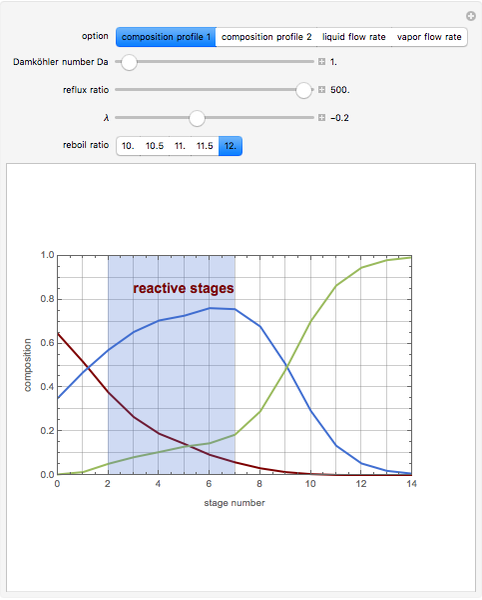
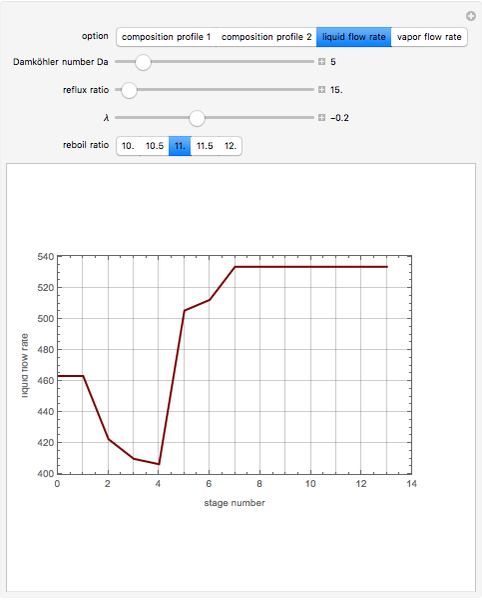
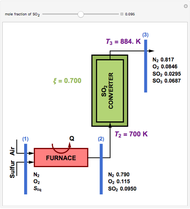
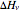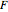The mixture is fed into a reactive distillation column with 14 total plates. The feed stage location is stage number 5, the reactive stages are from stage 2 to stage 7, and the feed composition is equimolar in  and
and  (i.e., the feed is composed of 40 mole%
(i.e., the feed is composed of 40 mole%  , 40 mole%
, 40 mole%  , and 20 mole%
, and 20 mole%  ). The feed flow rate is chosen as 100 kmol/hr.
). The feed flow rate is chosen as 100 kmol/hr.
Heat effects are included in the computation through the use of the negative dimensionless ratio,  where
where  and
and 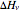 are the negative heat of reaction and the heat of vaporization, respectively (both expressed in kJ/kmol). One recovers the no-heat effects case by setting
are the negative heat of reaction and the heat of vaporization, respectively (both expressed in kJ/kmol). One recovers the no-heat effects case by setting  .
.
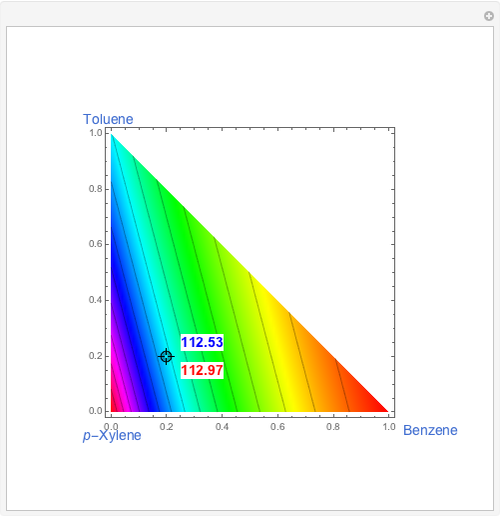
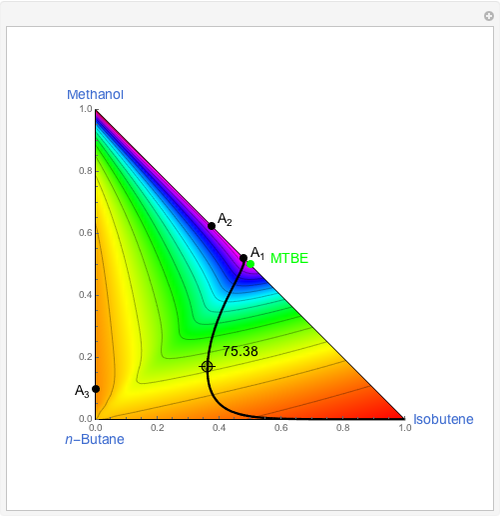
This Demonstration shows two profiles: (1) the composition versus plate number for components  ,
,  , and
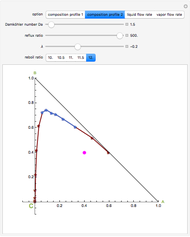
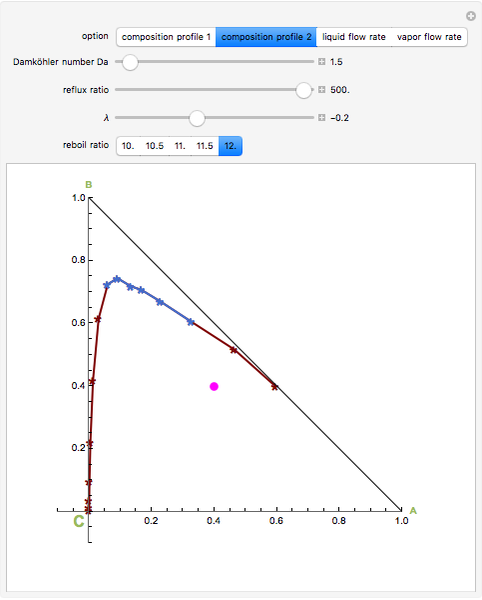
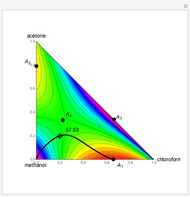
, and  (in red, blue, and green, respectively), with the reactive zone shown in light blue, and (2) a ternary diagram where the composition of
(in red, blue, and green, respectively), with the reactive zone shown in light blue, and (2) a ternary diagram where the composition of  versus the composition of
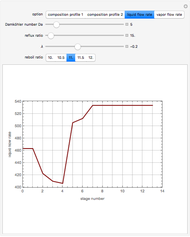
versus the composition of  is in mole%. In the second profile, the feed composition is shown by a magenta dot and the reactive stages (stages 2 to 7) are displayed in blue. The Demonstration also gives the nonconstant vapor and liquid flow rates in all stages. These flow rates obey the following relations, which are derived from global material and energy balances:
is in mole%. In the second profile, the feed composition is shown by a magenta dot and the reactive stages (stages 2 to 7) are displayed in blue. The Demonstration also gives the nonconstant vapor and liquid flow rates in all stages. These flow rates obey the following relations, which are derived from global material and energy balances:
 where
where  ,
,
 .
.
You can set the values of the Damköhler number,  , the reflux ratio, and the reboil ratio, where
, the reflux ratio, and the reboil ratio, where  is the plate molar hold-up,
is the plate molar hold-up, 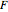 is the feed flow rate, and
is the feed flow rate, and  the reaction rate constant. When
the reaction rate constant. When  , one recovers the case where no reaction is taking place. If
, one recovers the case where no reaction is taking place. If  is very large, the simulation represents a situation close to reaction equilibrium (i.e.,
is very large, the simulation represents a situation close to reaction equilibrium (i.e.,  ). The Damköhler number is the ratio of the characteristic residence time to the characteristic reaction time.
). The Damköhler number is the ratio of the characteristic residence time to the characteristic reaction time.
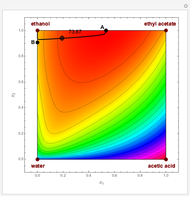
One snapshot shows a case where the reflux ratio is very high, a situation close to the total reflux operation. For such a situation, the bottom is almost pure  and the distillate contains very little
and the distillate contains very little  . The distillation flow is very small and the distillate stream can be recycled if desired. Thus, for this specific operation, one can produce
. The distillation flow is very small and the distillate stream can be recycled if desired. Thus, for this specific operation, one can produce  , if this is the desired reaction product, and convert both reactants
, if this is the desired reaction product, and convert both reactants  and
and  with a single piece of equipment.
with a single piece of equipment.
[less]

 ,
,  , and
, and  with relative volatilities
with relative volatilities  and
and  . This mixture is subject to an exothermic equilibrium-limited chemical reaction
. This mixture is subject to an exothermic equilibrium-limited chemical reaction  with reaction rate
with reaction rate  , where the equilibrium constant
, where the equilibrium constant  .
.