Separation of Methanol from Water by Batch Rectification

Requires a Wolfram Notebook System
Interact on desktop, mobile and cloud with the free Wolfram Player or other Wolfram Language products.
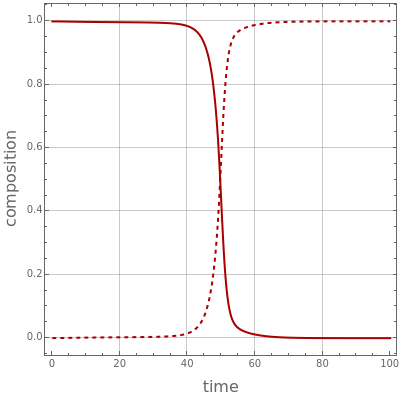
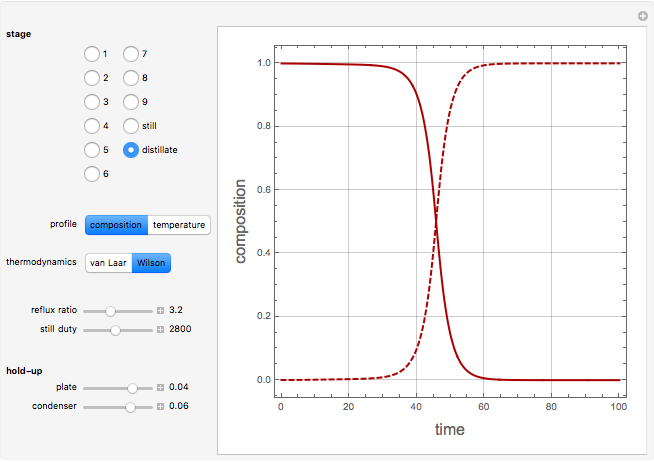
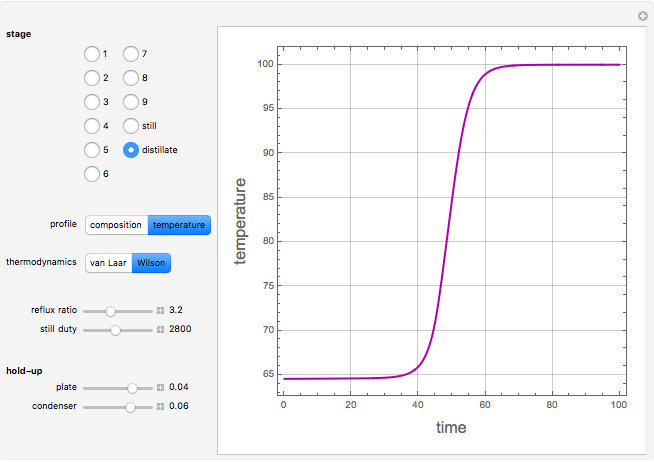
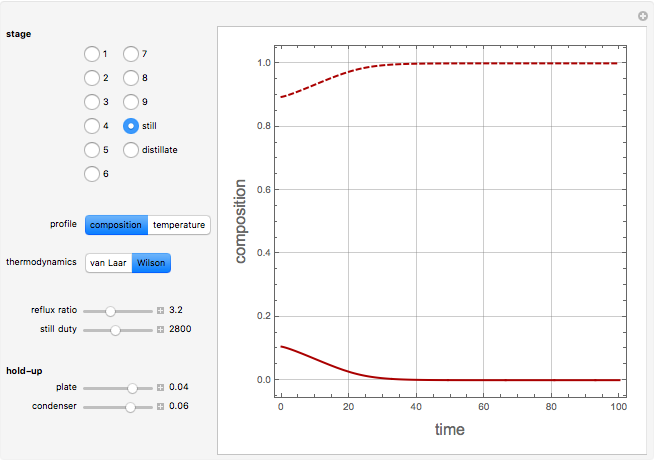
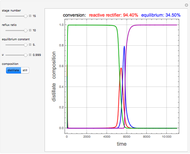
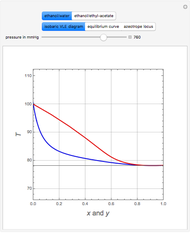
Consider a binary mixture containing 20 mol% methanol and 80 mol% water. This mixture is to be separated using a batch rectifier operating at  mmHg and composed of a still, nine trays, and a total condenser. You can set the values of the heating duty, the reflux ratio, the plateaus, and the condenser hold-ups. The vapor-liquid equilibria calculations are based on either the van Laar or Wilson models. Here, it is found that the choice of the thermodynamic model (to predict the activity coefficients) has very little effect on the composition and temperature profiles.
mmHg and composed of a still, nine trays, and a total condenser. You can set the values of the heating duty, the reflux ratio, the plateaus, and the condenser hold-ups. The vapor-liquid equilibria calculations are based on either the van Laar or Wilson models. Here, it is found that the choice of the thermodynamic model (to predict the activity coefficients) has very little effect on the composition and temperature profiles.
Contributed by: Housam Binous, Mamdouh Al-Harthi, and Ahmed Bellagi (December 2015)
Open content licensed under CC BY-NC-SA
Snapshots
Details
Reference
[1] U. Diwekar, Batch Distillation: Simulation, Optimal Design, and Control, 2nd ed., Boca Raton: CRC Press, 2012.
Permanent Citation
"Separation of Methanol from Water by Batch Rectification"
http://demonstrations.wolfram.com/SeparationOfMethanolFromWaterByBatchRectification/
Wolfram Demonstrations Project
Published: December 7 2015