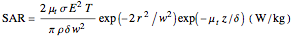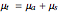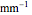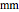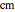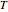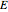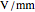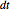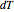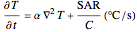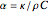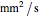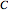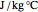Thermal Distribution Model of THz Radiation Absorption in Biological Tissue

Requires a Wolfram Notebook System
Interact on desktop, mobile and cloud with the free Wolfram Player or other Wolfram Language products.
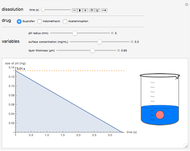
Terahertz radiation falls between the infrared and microwave regions of the electromagnetic spectrum, and it shares some properties with each of these. This Demonstration shows a closed-form thermal-change model due to THz continuous wave (CW) radiation regime absorption in biological tissue at room temperature. It uses the modified heat conduction equation, which assumes no heat response of body tissue homeostatic thermoregulation or convective or radiative heat transfer. Considering a medical application of THz radiation regime absorption in biological tissue, the model involves important parameters such as power density, the calculated beam waist, distance from power source, fraction of transmitted power, penetration depth of THz radiation, scattering and absorption coefficient, all of which are extremely important regarding heat generation due to irradiation on any biological tissue. The amount of heat inside the tissue is highly dependent on its optical properties, such as the absorption and scattering coefficients. Also, the heat transfer depends on some properties of biological tissues, such as thermal diffusivity, thermal and electric conductivity, and so on. Knowledge of some basic concepts of thermodynamics and heat transfer are necessary to understand and to evaluate the effects of THz radiation regime absorption on biological tissues for treatment purposes. You can set the tissue variables, optical parameters, and THz source variables. Vary the THz beam waist radius 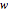 from sharper to broader or otherwise to see the significant change of temperature against depth of tissue.
from sharper to broader or otherwise to see the significant change of temperature against depth of tissue.
Contributed by: Muhamad Hamdi, Ahmad Zulhilmi bin Arshad, and Yusof Munajat (October 2013)
(University of Riau Indonesia, Pekanbaru, and University Technology of Malaysia, Johor Bahru)
Open content licensed under CC BY-NC-SA
Snapshots
Details
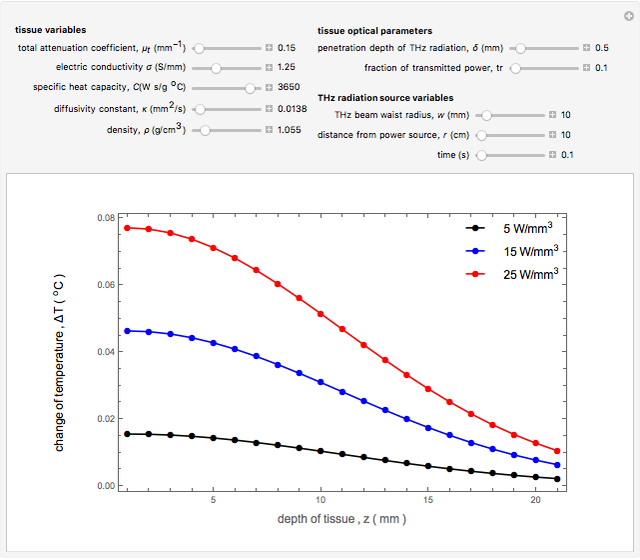
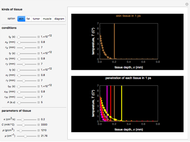
Snapshot 1: The model shows that the heat generated is directly related to the THz radiation regime absorption strength. THz radiation heat transfer of three different power densities shows that the heat changes against the depth of tumor tissue for the exposure with THz beam waist radius 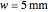 in
in 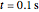 . The low power density (black) results in a small temperature change with increasing tissue depth and vice-versa (red and blue). For that, it is convenient to remember how the heat propagation occurs. When a THz radiation beam irradiates a tissue, the beam is partially reflected and the rest penetrates the tissue boundaries.
. The low power density (black) results in a small temperature change with increasing tissue depth and vice-versa (red and blue). For that, it is convenient to remember how the heat propagation occurs. When a THz radiation beam irradiates a tissue, the beam is partially reflected and the rest penetrates the tissue boundaries.
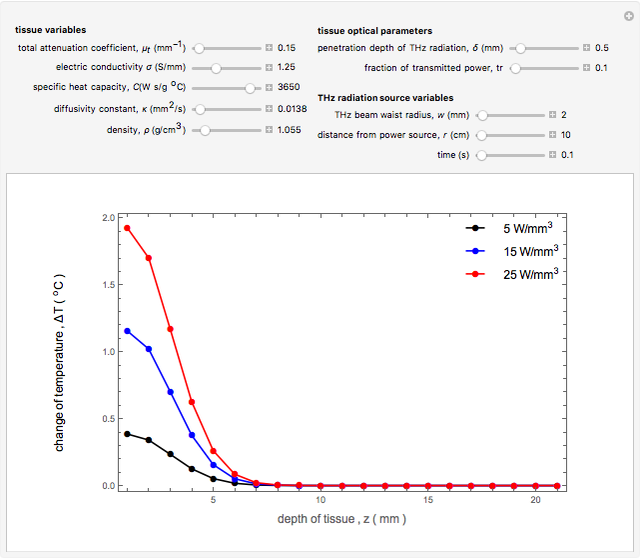
Snapshot 2 and 3: An increase of the wider THz beam waist radius from 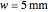 to
to 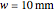 and
and 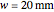 appears to decrease the temperature at time
appears to decrease the temperature at time 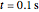 . The high (red) and medium (blue) power density results in a high temperature change against depth of tumor tissue, whereas the low power density (black) shows the slight temperature change to remain constant gradually.
. The high (red) and medium (blue) power density results in a high temperature change against depth of tumor tissue, whereas the low power density (black) shows the slight temperature change to remain constant gradually.
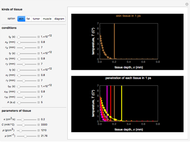
Snapshot 4: The change of the narrower THz beam waist radius from 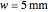 to
to 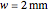 gives rise to a sharp increase (red and blue) in the temperature against a depth of tumor tissue about
gives rise to a sharp increase (red and blue) in the temperature against a depth of tumor tissue about 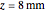 at time
at time 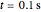 .
.
Phenomena of THz radiation absorption in biological tissue describe the fraction of the radiation after attenuation by absorption and scattering, provided that this penetrates the tissue. The absorption and the scattering are characterized by absorption coefficient 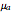 and scattering coefficient
and scattering coefficient 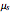 , which represent, respectively, the rate of radiation heat loss per penetration length unit, due to the absorption and the photons scattering. These two coefficients are specific to each tissue and depend on the THz radiation regime power density. The inverse of the absorption coefficient is defined as the optical absorption length. This represents the generated heat per area unit and per time unit in a very small thickness
, which represent, respectively, the rate of radiation heat loss per penetration length unit, due to the absorption and the photons scattering. These two coefficients are specific to each tissue and depend on the THz radiation regime power density. The inverse of the absorption coefficient is defined as the optical absorption length. This represents the generated heat per area unit and per time unit in a very small thickness 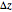 . This result can help to better understand the temperature history of living tissues subject to step heating and thus suggest requirements for the tumor-killing temperatures. Therefore the present investigation is more capable of dealing with many practical bioheat transfer problems than some other suggested analytical solutions.
. This result can help to better understand the temperature history of living tissues subject to step heating and thus suggest requirements for the tumor-killing temperatures. Therefore the present investigation is more capable of dealing with many practical bioheat transfer problems than some other suggested analytical solutions.
References
[1] M. Hamdi, Y. Munajat, R. K. R. Ibrahim, and R. A. Rahman, "Terahertz Radiation Field Regime Absorption in Cancer-Health Tissue for Medical Application," presentation given at The 4th International Conference & Workshop on Basic and Applied Science (2013), Johor Bahru, Malaysia.
[2] M. Hamdi, "Investigation on Bio-Electromagnetic Field of Terahertz Radiation Behaviours at Interface of Brain-Fat Tissue," Journal of Innovative Science, 1(2), 2012 p. 5.
Permanent Citation