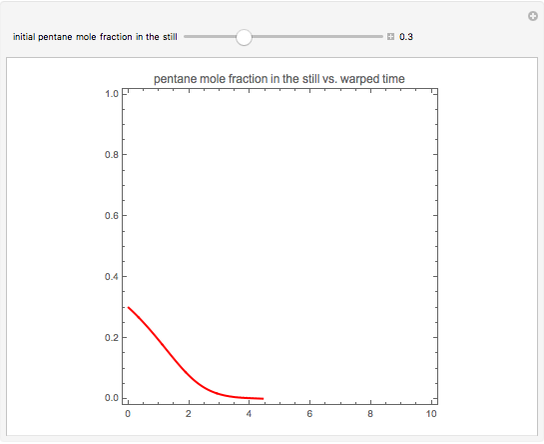
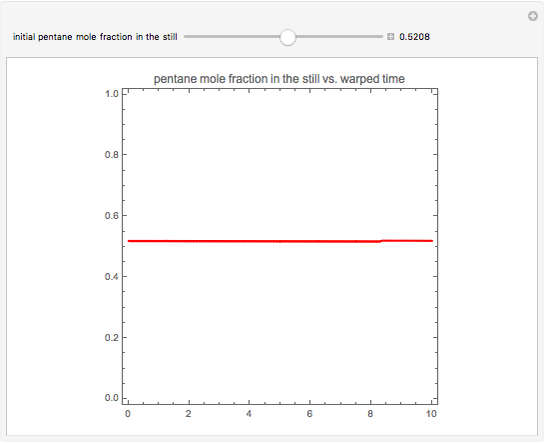
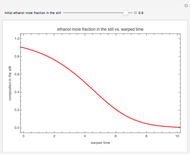
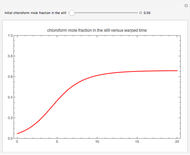
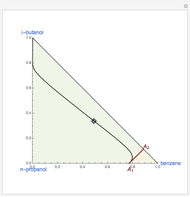
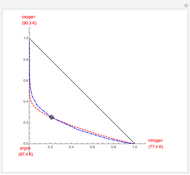
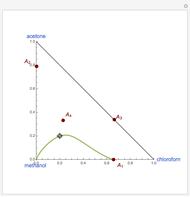
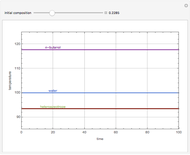
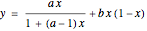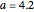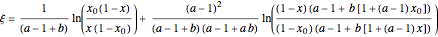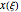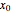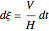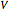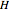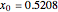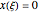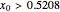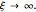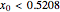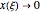Simple Distillation Residue Curves for a Mixture of Pentane and Dichloromethane at 750 mmHg
Initializing live version

Requires a Wolfram Notebook System
Interact on desktop, mobile and cloud with the free Wolfram Player or other Wolfram Language products.
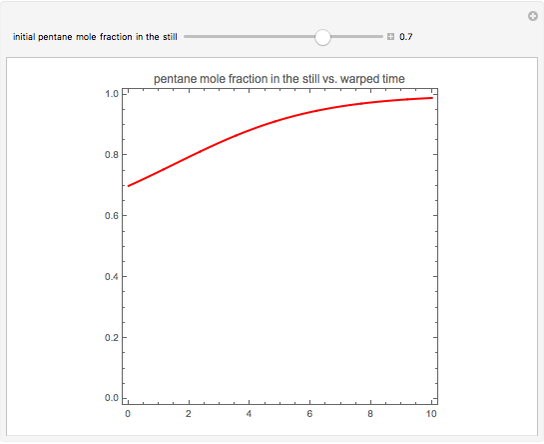
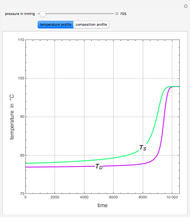
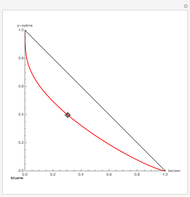
The vapor-liquid equilibrium behavior of the binary mixture composed of pentane and dichloromethane at 750 mmHg can be represented by the following empirical equation:
[more]
Contributed by: Housam Binous (March 2011)
Open content licensed under CC BY-NC-SA
Snapshots
Details
For more information, see:
M. F. Doherty and M. F. Malone, Conceptual Design of Distillation Systems, New York: McGraw-Hill, 2001.
Permanent Citation