Adiabatic Compression of Pure Gases

Requires a Wolfram Notebook System
Interact on desktop, mobile and cloud with the free Wolfram Player or other Wolfram Language products.
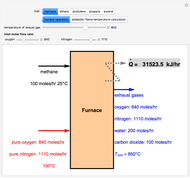
In this Demonstration,  -butane is compressed adiabatically from 1 bar and 50 °C to user-set values for the discharge stream pressure and the compressor efficiency
-butane is compressed adiabatically from 1 bar and 50 °C to user-set values for the discharge stream pressure and the compressor efficiency  . Click the "sketch of apparatus" button to determine the outlet stream temperature (expressed in °C) and the power consumed (expressed in kW). The inlet molar flow rate of
. Click the "sketch of apparatus" button to determine the outlet stream temperature (expressed in °C) and the power consumed (expressed in kW). The inlet molar flow rate of  -butane supplied to the compressor is
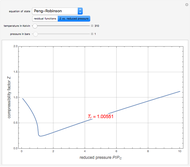
-butane supplied to the compressor is  . You can select from one of three cubic equations of state (Redlich–Kwong, Soave–Redlich–Kwong (SRK), or Peng–Robinson). An ideal gas at 1 bar and 298.15 K is taken as the reference state for computing the enthalpy
. You can select from one of three cubic equations of state (Redlich–Kwong, Soave–Redlich–Kwong (SRK), or Peng–Robinson). An ideal gas at 1 bar and 298.15 K is taken as the reference state for computing the enthalpy  and entropy
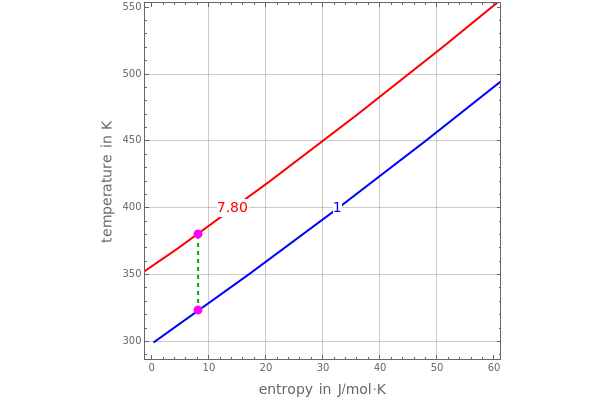
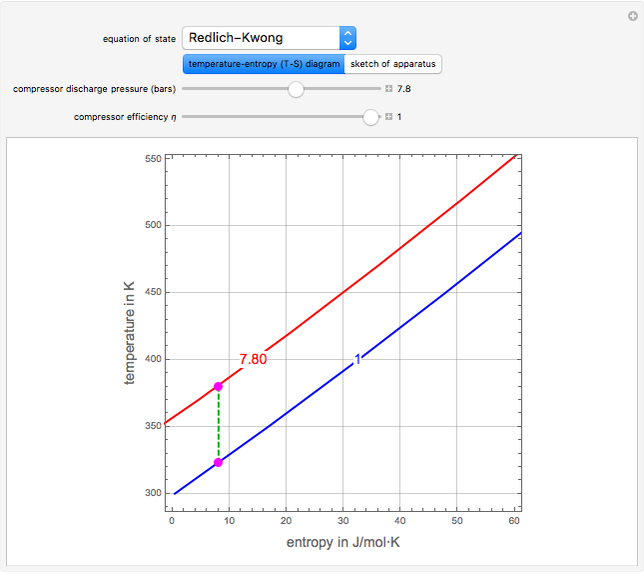
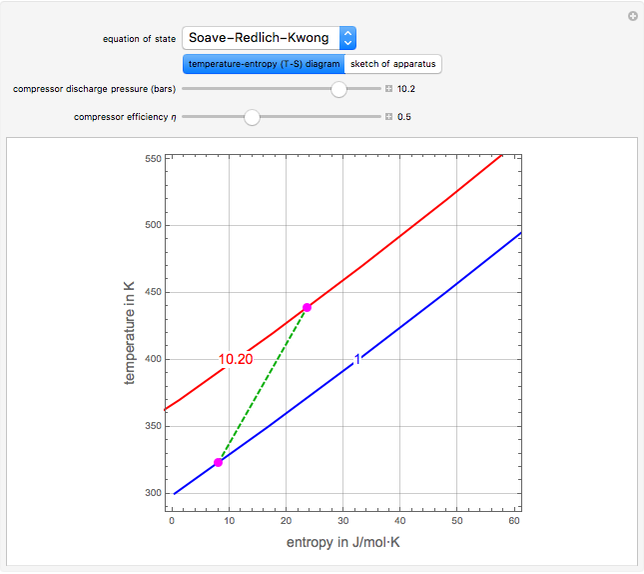
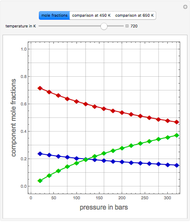
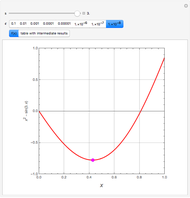
and entropy  for the system flow streams. In the temperature-entropy (T-S) diagram, the blue curve denotes the inlet stream isobar (
for the system flow streams. In the temperature-entropy (T-S) diagram, the blue curve denotes the inlet stream isobar ( ) and the red curve the discharge stream isobar. A limiting compression transformation on the T-S diagram is shown in dashed green. You can clearly see the difference between the ideal case (shown by a vertical dashed green line), when the compressor is isentropic (i.e.,
) and the red curve the discharge stream isobar. A limiting compression transformation on the T-S diagram is shown in dashed green. You can clearly see the difference between the ideal case (shown by a vertical dashed green line), when the compressor is isentropic (i.e.,  ), and a more realistic case, where the efficiency is less than 100% (shown by a tilted dashed green line).
), and a more realistic case, where the efficiency is less than 100% (shown by a tilted dashed green line).
Contributed by: Housam Binous, Nadhir A. Al-Baghli, and Brian G. Higgins (February 2012)
Open content licensed under CC BY-NC-SA
Snapshots
Details
Results obtained by this Demonstration are in excellent agreement with values obtained by a process simulator, HYSYS, for the Peng–Robinson and SRK equations of state. Compression of other pure gases can be studied using the program here simply by changing the critical temperature,  , and pressure,
, and pressure,  , as well as the acentric factor,
, as well as the acentric factor,  .
.
Reference
[1] J. M. Smith, H. C. Van Ness, and M. M. Abbott, Introduction to Chemical Engineering Thermodynamics, 7th ed. (international edition), New York: McGraw-Hill, 2005.
Permanent Citation