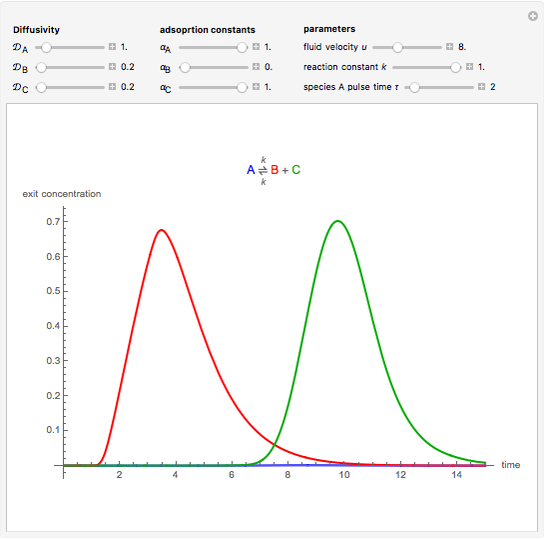
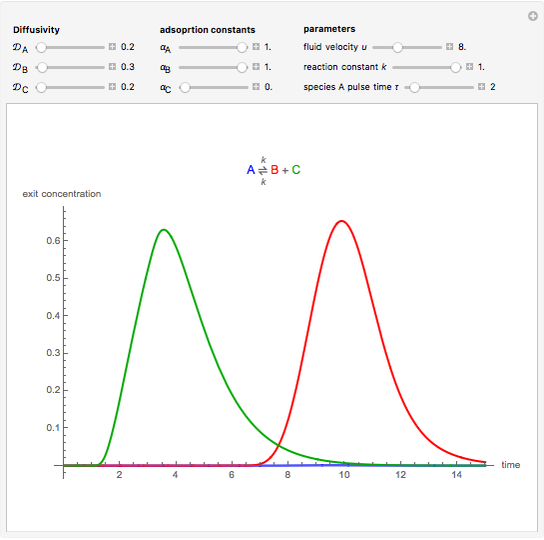
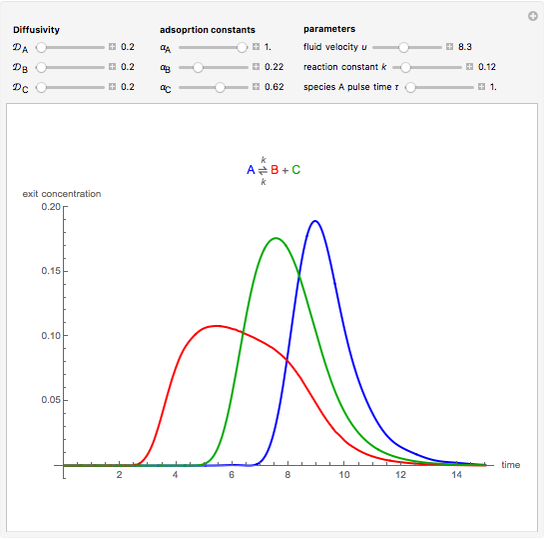
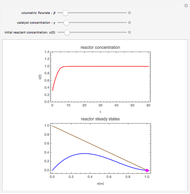
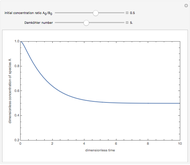
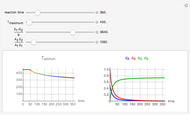
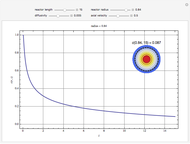
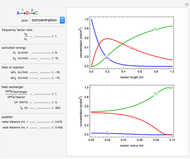
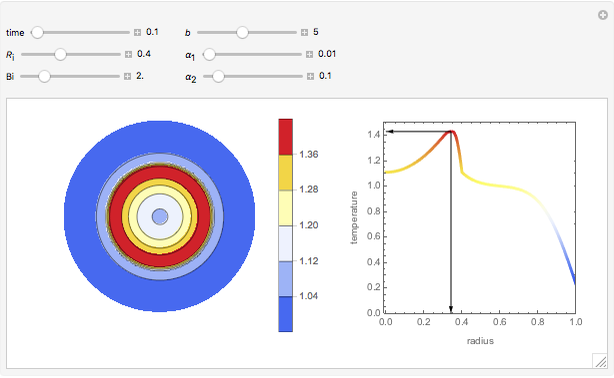
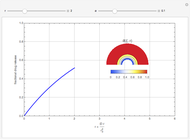
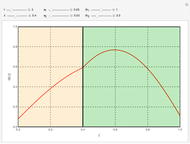
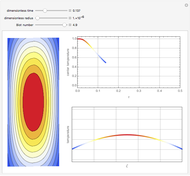
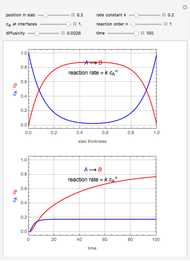
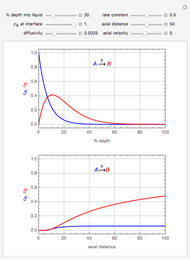
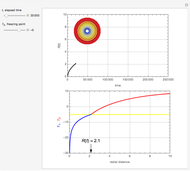
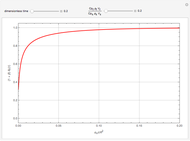
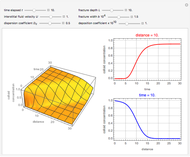
Chromatographic reactors integrate chemical reaction and separation in one apparatus. This Demonstration analyses the reaction  conducted in a chromatograph.
conducted in a chromatograph.
The equations describing the fluid concentration  of the three species in a fixed-bed chromatography column are:
of the three species in a fixed-bed chromatography column are:
 ,
,
where  is time,
is time,  is distance,
is distance,  represents porosity,
represents porosity,  stands for the effective diffusivity,
stands for the effective diffusivity,  is the rate of chemical reaction (negative for reactants and positive for products), and the
is the rate of chemical reaction (negative for reactants and positive for products), and the  are the solid-phase concentrations that are related to the fluid concentrations via adsorption constants
are the solid-phase concentrations that are related to the fluid concentrations via adsorption constants  ,
,  .
.
The reaction rate is  , and the initial and boundary conditions are:
, and the initial and boundary conditions are:
 ,
,
 ,
,
 , and
, and
 ,
,  ,
where
,
where  is the time that species
is the time that species  is injected into the chromatograph. Chromatographic separation is based primarily on the difference in adsorptivity
is injected into the chromatograph. Chromatographic separation is based primarily on the difference in adsorptivity  , as well as the difference in the diffusion coefficient
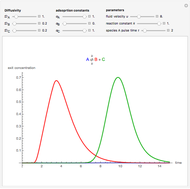
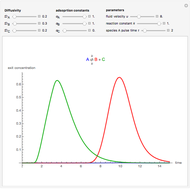
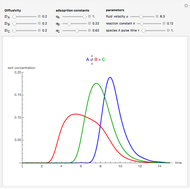
, as well as the difference in the diffusion coefficient  You can follow the trajectory of the system by varying adsorptivities, diffusion coefficients, fluid velocity, reaction rate, and the time of injection of reactant
You can follow the trajectory of the system by varying adsorptivities, diffusion coefficients, fluid velocity, reaction rate, and the time of injection of reactant  .
.
[less]

 conducted in a chromatograph.
conducted in a chromatograph.