Saturation Binding of Ligands to Proteins

Requires a Wolfram Notebook System
Interact on desktop, mobile and cloud with the free Wolfram Player or other Wolfram Language products.
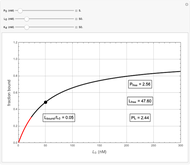
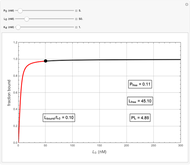
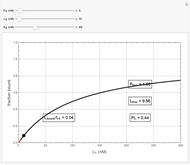
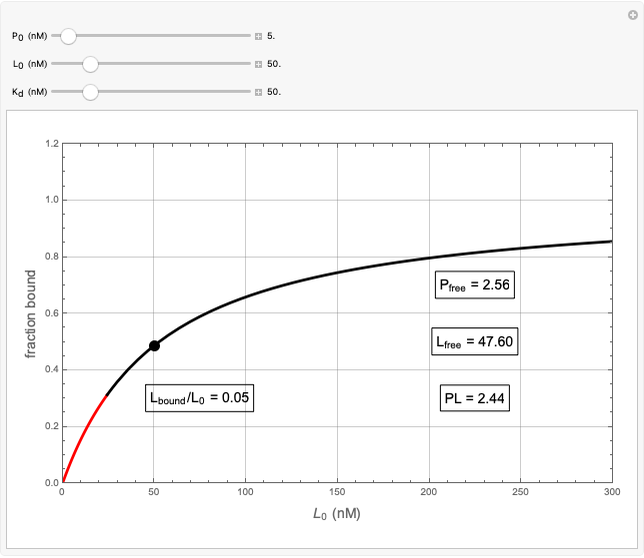
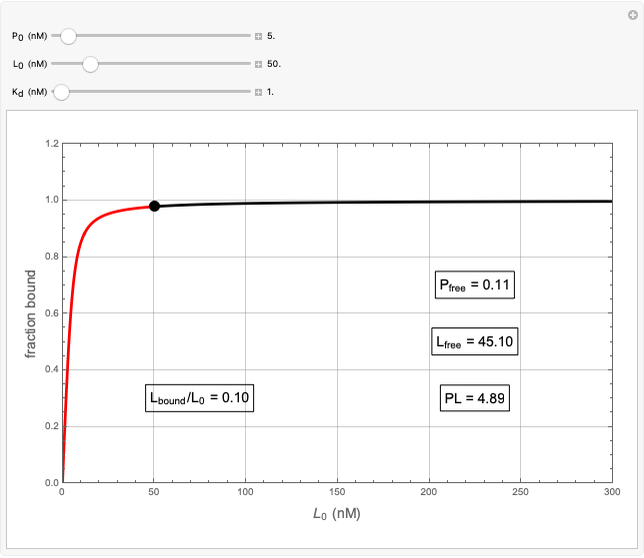
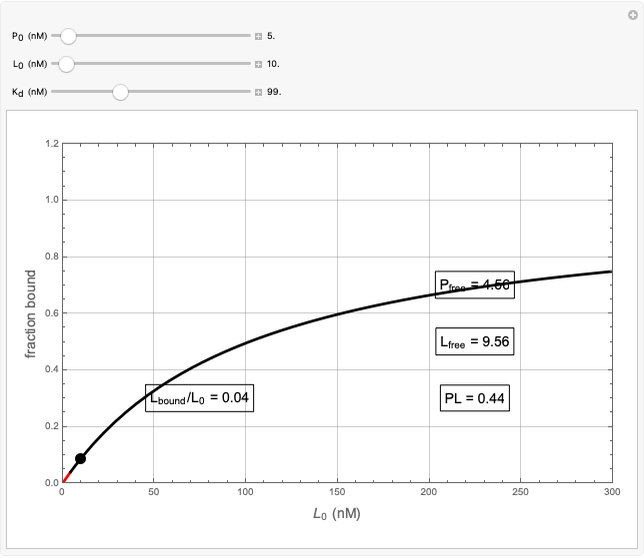
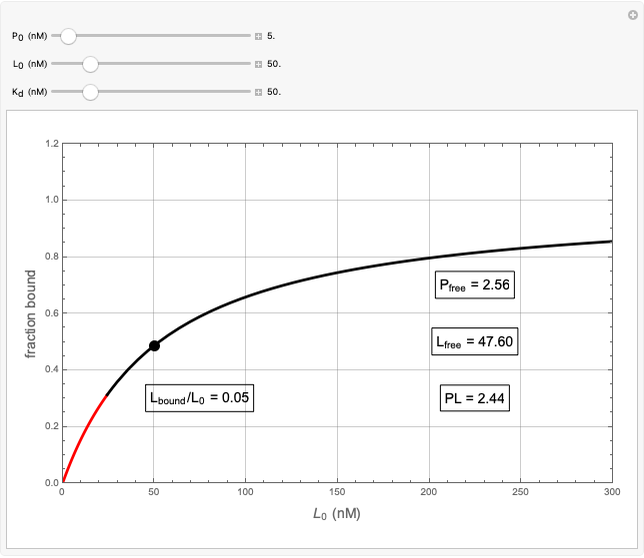
Basic saturation binding experiments to determine dissociation constants  ) assume that the concentration of free ligands is about the same (greater than 90%) as the concentration of ligands added. If not, the results could be invalid due to ligand depletion. A general rule of thumb is to use a starting concentration of protein
) assume that the concentration of free ligands is about the same (greater than 90%) as the concentration of ligands added. If not, the results could be invalid due to ligand depletion. A general rule of thumb is to use a starting concentration of protein  well below (~10 times) the estimated
well below (~10 times) the estimated  , and ligand concentrations
, and ligand concentrations  that bracket the
that bracket the  by at least 10 times on either side. Many published experiments ignore this rule. This graphical aid simplifies experimental design by readily showing safe macromolecule/ligand concentrations in black and dangerous conditions in red.
by at least 10 times on either side. Many published experiments ignore this rule. This graphical aid simplifies experimental design by readily showing safe macromolecule/ligand concentrations in black and dangerous conditions in red.  ,
,  , and
, and  all have the same units of concentration (nM in this Demonstration).
all have the same units of concentration (nM in this Demonstration).
Contributed by: Karl Volz (February 2012)
Open content licensed under CC BY-NC-SA
Details
The equation used is

References
[1] K. B. Hall and J. K. Kranz, "Nitrocellulose Filter Binding for Determination of Dissociation Constants," Methods in Molecular Biology, 118, 1999 pp. 105–114.
[2] H. Motulsky, "GraphPad Prism Version 4.0 Statistics Guide," La Jolla, CA: GraphPad Software, Inc., 2005.
Snapshots
Permanent Citation