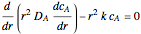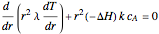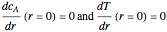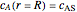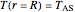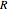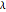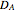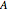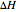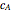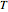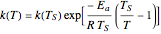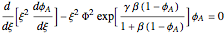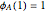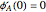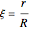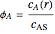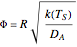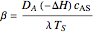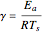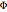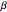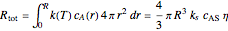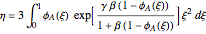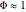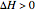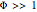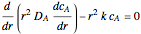 ,
,
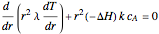 ,
,
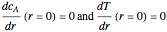 ,
,
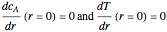 ,
,
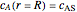 and
and 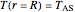 ,
,
where 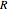 is the radius of the spherical pellet,
is the radius of the spherical pellet, 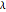 is the effective thermal conductivity of the pellet,
is the effective thermal conductivity of the pellet,  the reaction rate constant,
the reaction rate constant, 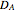 the effective binary diffusivity of
the effective binary diffusivity of 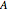 within the pellet,
within the pellet, 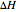 the heat of reaction,
the heat of reaction, 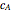 the concentration, and
the concentration, and 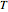 the temperature.
the temperature.
The rate constant is a function of temperature: 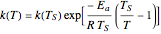 .
.
In terms of dimensionless quantities, we have the following boundary value problem:
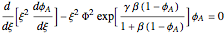 with
with 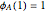 and
and 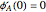 ,
,
where 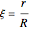 and
and 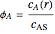 .
.
Three dimensionless parameters are introduced in the new governing equation:
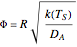 ,
, 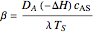 and
and 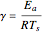 , where
, where 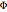 , the Thiele modulus, is a measure of internal mass transfer resistance. When
, the Thiele modulus, is a measure of internal mass transfer resistance. When 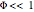 , mass transfer resistance is negligible and diffusion is very fast. Large values of
, mass transfer resistance is negligible and diffusion is very fast. Large values of  , the dimensionless activation energy, mean that the reaction rate is very sensitive to temperature. In this Demonstration
, the dimensionless activation energy, mean that the reaction rate is very sensitive to temperature. In this Demonstration  is taken equal to 1.
is taken equal to 1.
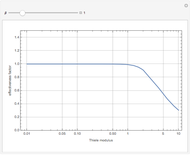
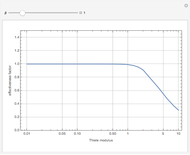
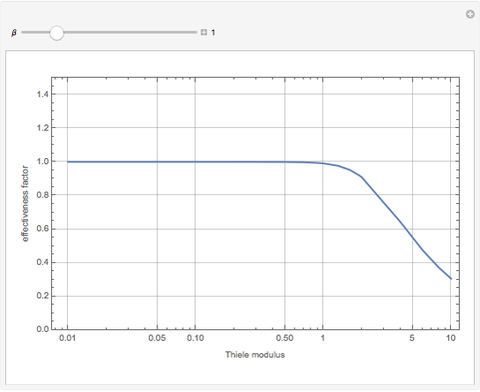
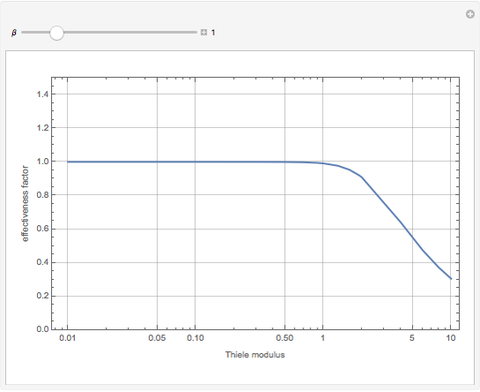
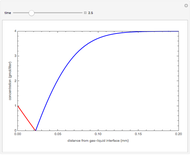
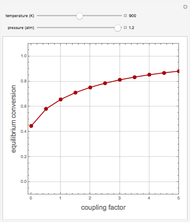
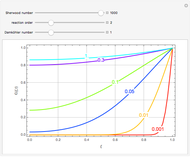
The Demonstration displays the effectiveness factor,  , a measure of the importance of the total reaction rate inside the pellet compared to its value at the pellet surface, versus the Thiele modulus
, a measure of the importance of the total reaction rate inside the pellet compared to its value at the pellet surface, versus the Thiele modulus 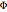 , for various values of parameter
, for various values of parameter 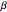 .
.
The effectiveness factor is given by the formula:
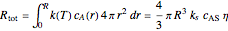 ,
,
where 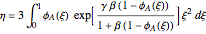 .
.
The parameter 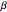 , which you can set, is a measure of the relative importance of the heat of reaction relative to conduction.
, which you can set, is a measure of the relative importance of the heat of reaction relative to conduction.
Large values of 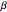 mean that there is significant internal heating; thus for
mean that there is significant internal heating; thus for 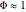 the effectiveness factor will be greater than 1. This means that the rate of reaction is higher inside the pellet compared to its value at the surface of the pellet in the absence of internal diffusion resistance.
the effectiveness factor will be greater than 1. This means that the rate of reaction is higher inside the pellet compared to its value at the surface of the pellet in the absence of internal diffusion resistance.
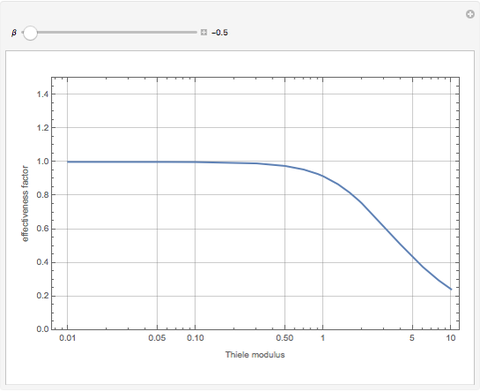
Negative values of the parameter 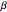 correspond to endothermic reactions (
correspond to endothermic reactions (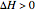 ) and smaller values of the effectiveness factor,
) and smaller values of the effectiveness factor,  .
.
When 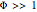 , the internal transport resistance becomes large (a depleted zone inside the pellet forms and the concentration of species
, the internal transport resistance becomes large (a depleted zone inside the pellet forms and the concentration of species 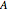 is low). Thus, the reaction rate inside the pellet is smaller than its value at the pellet surface and consequently the effectiveness factor becomes very small compared to 1.
is low). Thus, the reaction rate inside the pellet is smaller than its value at the pellet surface and consequently the effectiveness factor becomes very small compared to 1.
[less]

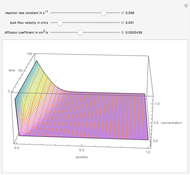
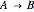 taking place in a spherical catalyst pellet. The governing equations, obtained from mole and enthalpy balances, and the boundary conditions, obtained from the symmetry condition and the known values at the surface, are the following:
taking place in a spherical catalyst pellet. The governing equations, obtained from mole and enthalpy balances, and the boundary conditions, obtained from the symmetry condition and the known values at the surface, are the following: