Sensitivity Analysis of Transition Phases of Perturbed Gene Pathways with a Neural Network

Requires a Wolfram Notebook System
Interact on desktop, mobile and cloud with the free Wolfram Player or other Wolfram Language products.
This Demonstration shows for the first time, a sensitivity analysis method based on an artificial neural network to find transition phases from homeostasis toward liver cirrhosis in hepatocytes infected by HCV. Currently, finding transition phases represents one of the fundamental steps in understanding tumor transformation. The Demonstration examines the cytokeratins gene expression profile, which was organized in a pathway by means of Ingenuity Pathway Analysis (IPA) software. Gene expression values were obtained from DNA microarrays of normal hepatocytes versus cirrhosis with HCV infection. The Demonstration describes a fast way of "handling" gene expression parameters that determine the transition from a healthy condition to cirrhosis within the cytokeratins context. This approach permits singling out critical "catastrophe points", simply perturbing an independent variable (such as a gene within the cytokeratins pathway) and visualizing how the perturbation impacts a particular dependent variable. This Demonstration could answer two fundamental questions: (1) how moving the control bar for a specific gene can generate the transition from normal condition to cirrhosis, and (2) moving more control bars, what minimal movement of a group of genes can produce the same perturbation. This method could be useful in measuring relationship fluxes among genes, which currently have not been described in the literature, suggesting new experimental designs.
Contributed by: Luca Zammataro (March 2011)
Open content licensed under CC BY-NC-SA
Snapshots
Details
Background
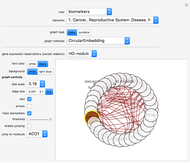
The program represents a biological application of an artificial neural network in the recently published Demonstration, "Solving the Hole in the Square Problem with a Neural Network".
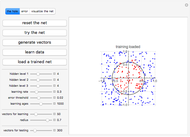
The neural network architecture used for this Demonstration is composed of 34 input neurons, one output neuron, and three hidden neuron levels, each with eight units. The net was trained with the expression values of 34 genes observed in two biological conditions, normal and liver cirrhosis. There were 22 patients consisting of 10 normal donors and 11 with cirrhosis. The gene expression dataset was obtained from DNA microarrays of normal hepatocytes versus cirrhosis with HCV infection. Gene expressions were filtered with a  -test (
-test (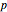 -values less than 0.01), and with fold change greater than
-values less than 0.01), and with fold change greater than  and less than
and less than 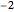 .
.
The neural network was trained with a training set of patterns (when this Demonstration was written), which were obtained transforming the gene value within the cytokeratins network in a series of sample patterns, each one corresponding to a gene expression dataset of a donor. We can call this set of patterns a training set because it is composed of the 22 patterns. Each pattern is composed of 34 parameters (gene expressions) and their expected outputs: 0 if the gene values correspond to normal conditions, and 1 if the gene values correspond to cirrhosis. Inside the neural weights, an association between phenotype and the 34 gene expression values occurs. The program uses this association to infer relationships among genes.
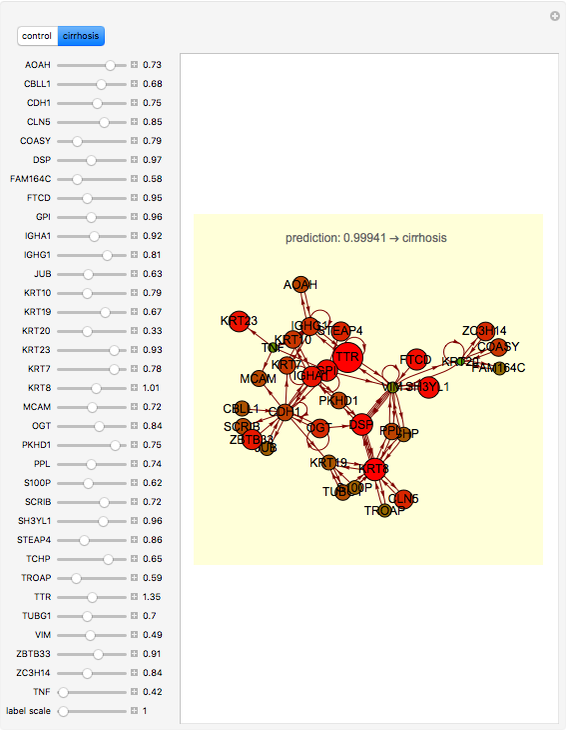
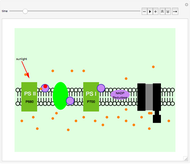
This neural network is not able to learn in this Demonstration. It can only compute parameter variations obtained by acting on the 34 control bars while the program is running. Click the "control" button to load the net with the cytokeratins gene profile in normal condition. Click the "cirrhosis" button to load the net with the cytokeratins gene profile in cirrhosis condition. Skipping from normal to cirrhosis, the net produces outputs associated with the two conditions: 0 for normal and 1 for cirrhosis. When the output produced is normal, the background becomes cyan; when the output is cirrhosis, the background is light yellow.
The method described here uses information from systems biology (interaction among molecules, obtained from literature) and data from DNA microarrays. All genes represented within the pathway were chosen from the microarrays by IPA, considering the significance, expressed as 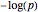 (using the
(using the  -value) based on a Fisher's exact test. In the case of the cytokeratins pathway, the significance was less than 0.01.
-value) based on a Fisher's exact test. In the case of the cytokeratins pathway, the significance was less than 0.01.
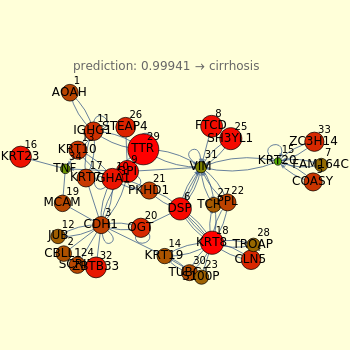
Moving the 34 bars, you can perturb the cytokeratins pathway. Each value is expressed as the log of the expression. Low values correspond to low expression of a gene, (downregulation). High values represent upregulations. The color and size of disks within the pathway represent the amount/decrease of gene transcription (expression). Large red disks correspond to upregulated genes; small green disks correspond to downregulated genes.
Playing with Genes
An amusing exercise is to move the bars and to try to understand what relationships govern the fate of the pathway. An example is given by the unclear role of KRT23, which is responsible for a fast transition from normal to cirrhosis; you can demonstrate this moving the KRT23 gene bar versus upregulation. The neural network attributes the increase of KRT23 to cirrhosis, and this depends on the gene expression of cirrhosis in comparison with the normal conditions used to train the neural network.
When you vary the S100P, the neural network indicates that S100P is one of the parameters that "has a weight" in contrasting the KRT23 increases. In fact, high levels of S100P together with high levels of KRT23 correspond to healthy conditions, as shown by the score in the top of the graphic. The neural network could be useful to highlight nonlinear relationships between two genes, such as S100P and KRT23, that are not well characterized experimentally.
The aim of the present Demonstration is to describe a computational method for sensitivity analysis and an artificial neural network that is able to reveal a transition phase between normal and cirrhotic equilibria, and at the same time to identify a circuitry that includes molecules such as TNF-alpha, citokeratine, and adhesion molecules and their regulators. A detailed explanation of these 34 molecules goes outside the scope of this Demonstration and requires knowledge of biology.
Playing with these 34 genes, a plethora of molecular actors emerge (CDH1, TNF, KRTs), and the existence of a regulation loop between cadherin (CDH) and cytokeratins regulation, in great part regulated by TNF-alpha, suggests the presence of one of the possible mechanisms in fibrosis/dysplasia/cirrhosis onset. Within this very complex circuitry there are molecules that interact with CDH1 and KRT19, some of these having a downregulation control: GPI protein decreases expression of human CDH1 protein while S100P protein is involved in the expression of human KRT19 protein. Considering the implications of cytokeratins metabolism in cell adhesion, cytoskeleton organization, and cell movement, the study of the transition phases represents the way to find the specific time at which, through a not well-known mechanism, the organizational capacity of the hepatocytes cytoskeleton appears destabilized, at the same time altering the cell contact mediated by adhesion molecules such as cadherins. This disordered condition results in the formation of non-ordered fibrous septa, which would be responsible for the beginning of the fibrotic process that leads to cirrhosis.
References
[1] T. Funasaka, V. Hogan, and A. Raz, "Phosphoglucose Isomerase/Autocrine Motility Factor Mediates Epithelial and Mesenchymal Phenotype Conversions in Breast Cancer," Cancer Research, 69(13), 2009 pp. 5349–5356.
[2] H. J. Whiteman, M. E. Weeks, S. E. Dowen, S. Barry, J. F. Timms, N. R. Lemoine, and T. Crnogorac–Jurcevic, "The Role of S100P in the Invasion of Pancreatic Cancer Cells Is Mediated through Cytoskeletal Changes and Regulation of Cathepsin D," Cancer Research, 67(18), 2007 pp. 8633–8642.
[3] M-Y. Hsu, F. E. Meier, M. Nesbit, J-Y. Hsu, P. Van Belle, D. E. Elder, and M. Herlyn, "E-Cadherin Expression in Melanoma Cells Restores Keratinocyte-Mediated Growth Control and Down-Regulates Expression of Invasion-Related Adhesion Receptors," American Journal of Pathology 156(5), 2000 pp. 1515–1525.
[4] C. Ren, F. Paronetto, K. M. Mak, M. A. Leo, and C. S. Lieber, "Cytokeratin 7 Staining of Hepatocytes Predicts Progression to More Severe Fibrosis in Alcohol-Fed Baboons," Journal of Hepatology, 38(6), 2003 pp. 770–775. PubMed PMID: 12763370.
[5] E. Wurmbach, et al., "Genome-Wide Molecular Profiles of HCV-Induced Dysplasia and Hepatocellular Carcinoma," Hepatology 45(4), 2007 pp. 938–947.
Permanent Citation