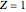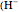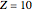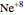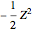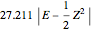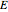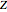Van Vleck's Model of the Helium Atom in the Old Quantum Theory

Requires a Wolfram Notebook System
Interact on desktop, mobile and cloud with the free Wolfram Player or other Wolfram Language products.
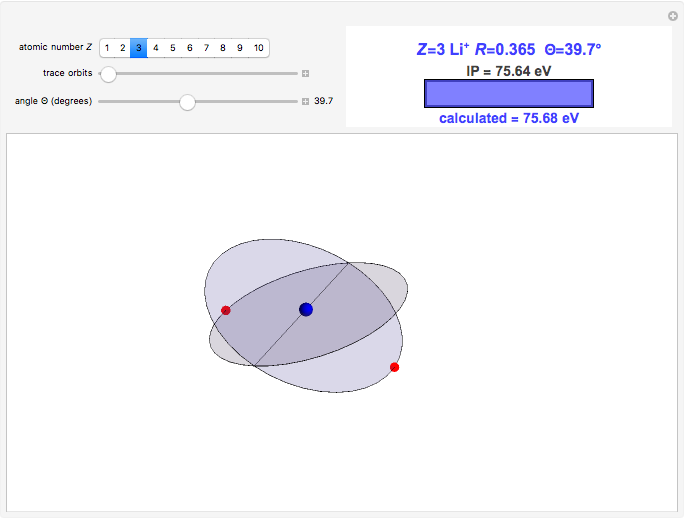
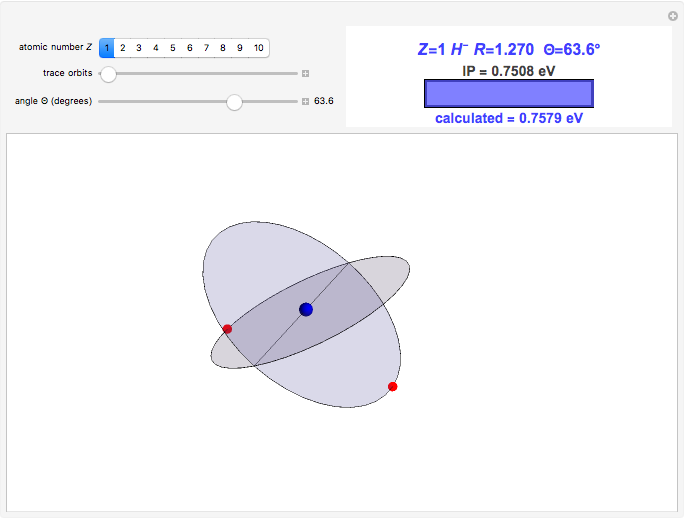
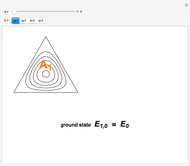
Niels Bohr's 1913 model of the hydrogen atom, now known as the old quantum theory (OQT), was a spectacular success in accounting for the spectrum of atomic hydrogen and introducing the concept of energy-level quantization. However, many valiant attempts to extend the Bohr model to the helium atom and beyond met with miserable failure, necessarily awaiting the development of quantum mechanics in 1925–1926. This Demonstration reproduces probably the most creditable OQT model of the helium atom, which was proposed in the doctoral dissertation of J. H. Van Vleck in 1922 [1, 2].
[more]
Contributed by: S. M. Blinder (December 2017)
Open content licensed under CC BY-NC-SA
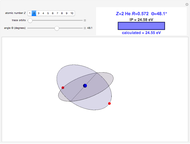
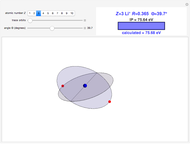
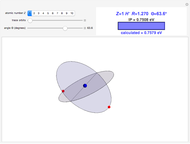
Snapshots
Details
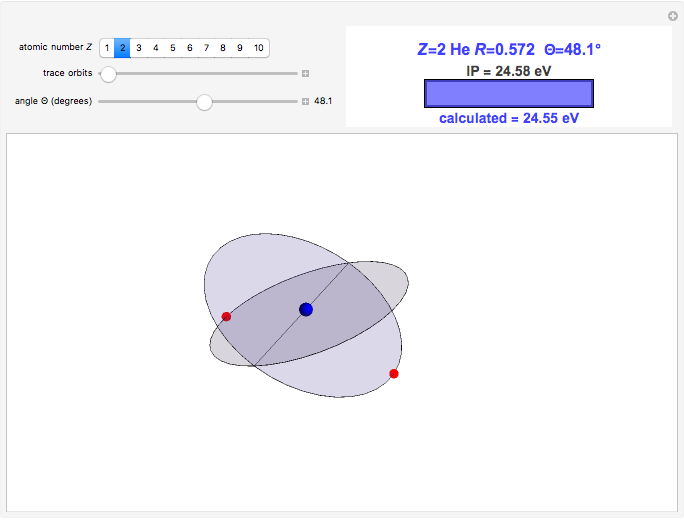
In this model for the motion of the two electrons in atomic helium, the orbits trace out ellipses representing two oblique cross-sections of a cylinder of radius  , mutually intersecting at an angle
, mutually intersecting at an angle  . The two electrons, in cylindrical coordinates, are specified by:
. The two electrons, in cylindrical coordinates, are specified by:
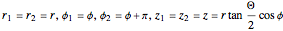 .
.
The kinetic energy is given by 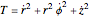 . Each electron-nuclear distance equals
. Each electron-nuclear distance equals 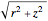 , while the electron-electron distance equals
, while the electron-electron distance equals 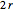 , thus giving a potential energy, in atomic units,
, thus giving a potential energy, in atomic units, 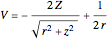 .
.
The Lagrangian
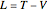
leads to the equations of motion:
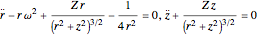 ,
, 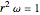 , where
, where 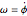 .
.
Earlier suggestions of a "crossed-orbit" model of the helium atom were made by Neils Bohr and his assistant, H. A. Kramers, as well as by E. C. Kemble, who was Van Vleck's doctoral advisor. The resulting dissertation is often considered the first American PhD thesis purely devoted to a topic in theoretical quantum physics. A personal note: Nobel laureate J. H. Van Vleck [3] was co-advisor for my own doctoral dissertation.
References
[1] J. H. Van Vleck, "A Critical Study of Possible Models of the Normal Helium Atom," PhD dissertation, Harvard University, 1922.
[2] J. H. Van Vleck, "The Normal Helium Atom and Its Relation to the Quantum Theory," Philosophical Magazine 44(263), 1924 pp. 842–869. doi.org/10.1080/14786441208562559.
[3] Wikipedia. "John Hasbrouck Van Vleck." (Dec 4, 2017) en.wikipedia.org/wiki/John_Hasbrouck_Van _Vleck.
Permanent Citation