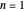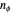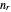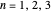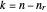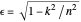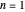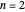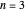The Fine-Structure Constant from the Old Quantum Theory

Requires a Wolfram Notebook System
Interact on desktop, mobile and cloud with the free Wolfram Player or other Wolfram Language products.
The fine structure constant, 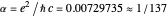 , measures the relative strength of the electromagnetic coupling constant in quantum field theory. Its small magnitude enables very accurate predictions in the perturbation expansions of quantum electrodynamics. This famous dimensionless parameter was first introduced by Arnold Sommerfeld in 1916 in a relativistic generalization of Bohr's atomic theory. As its simplest physical realization, the fine structure constant
, measures the relative strength of the electromagnetic coupling constant in quantum field theory. Its small magnitude enables very accurate predictions in the perturbation expansions of quantum electrodynamics. This famous dimensionless parameter was first introduced by Arnold Sommerfeld in 1916 in a relativistic generalization of Bohr's atomic theory. As its simplest physical realization, the fine structure constant 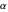 is equal to the ratio of the speed of the electron in the first Bohr orbit to the speed of light.
is equal to the ratio of the speed of the electron in the first Bohr orbit to the speed of light.
Contributed by: S. M. Blinder (March 2012)
Open content licensed under CC BY-NC-SA
Snapshots
Details
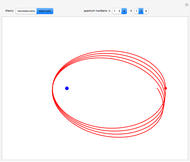
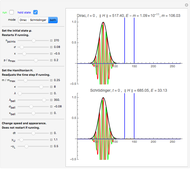
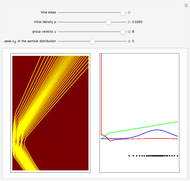
The nonrelativistic version of the old quantum theory was most succinctly expressed by the Sommerfeld–Wilson quantum conditions: 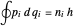 , for each periodic degree of freedom in the system. For example, the Keplerian orbital motion of a bound state of the electron in a hydrogen atom, an ellipse confined to a plane, has two degrees of freedom. The Hamiltonian can be written
, for each periodic degree of freedom in the system. For example, the Keplerian orbital motion of a bound state of the electron in a hydrogen atom, an ellipse confined to a plane, has two degrees of freedom. The Hamiltonian can be written 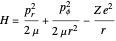 , where
, where 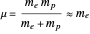 is the reduced mass and
is the reduced mass and 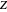 is the nuclear charge (
is the nuclear charge (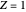 for
for 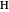 ,
, 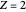 for
for 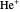 , etc.). For simplicity, we use atomic units
, etc.). For simplicity, we use atomic units 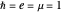 and assume infinite nuclear mass. Thus the Bohr radius is
and assume infinite nuclear mass. Thus the Bohr radius is 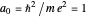 . The two quantum conditions are the phase integrals
. The two quantum conditions are the phase integrals 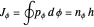 and
and 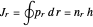 . The first is simple, since the Hamiltonian is independent of
. The first is simple, since the Hamiltonian is independent of  , so that
, so that 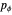 is a constant. Thus
is a constant. Thus 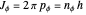 or
or 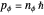 , with
, with 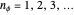 . This represents, in fact, the quantization of angular momentum. The integral
. This represents, in fact, the quantization of angular momentum. The integral 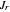 is a bit more challenging:
is a bit more challenging: 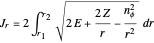 , where
, where 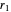 and
and 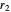 are the periapsis and apoapsis of the orbit. This works out to
are the periapsis and apoapsis of the orbit. This works out to 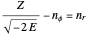 , with
, with 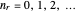 . The energy, in atomic units, reduces to the familiar Bohr formula
. The energy, in atomic units, reduces to the familiar Bohr formula 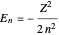 , where the principal quantum number
, where the principal quantum number 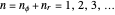 , and the azimuthal quantum number
, and the azimuthal quantum number 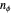 , usually written as
, usually written as  , has the allowed values
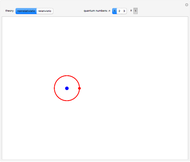
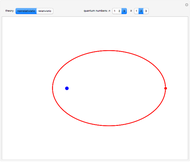
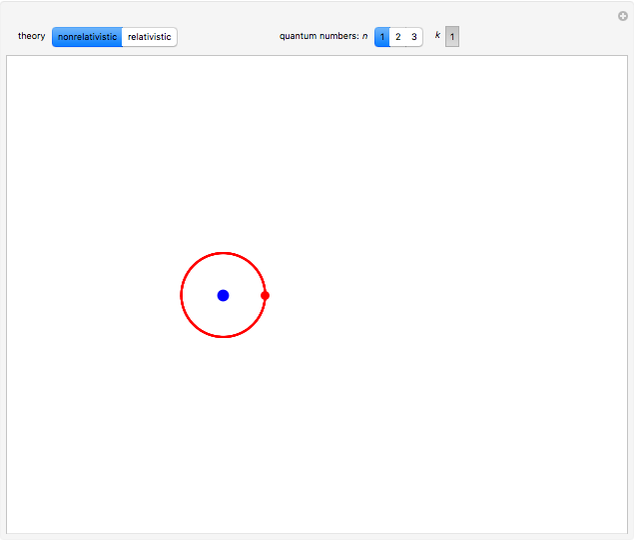
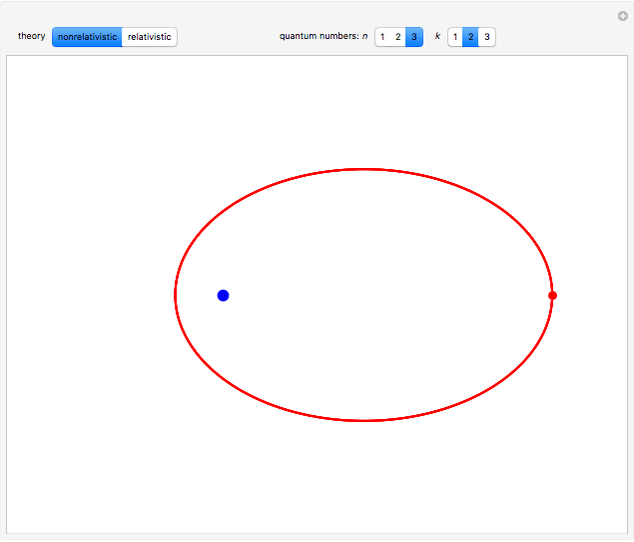
, has the allowed values 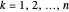 . The elliptical orbits are given by the polar equations
. The elliptical orbits are given by the polar equations 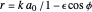 , with the eccentricity
, with the eccentricity 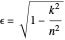 .
.
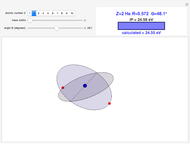
In the relativistic generalization of Sommerfeld's orbits, the radial and angular momenta of the electron are given by 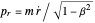 ,
, 
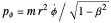 , where
, where 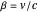 . These contain essentially relativistic generalizations of the components of kinetic energy. The Hamiltonian takes the form
. These contain essentially relativistic generalizations of the components of kinetic energy. The Hamiltonian takes the form 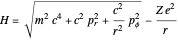 . Evaluation of the phase integrals is more complicated, and the interested reader is directed to the references. The relativistic energy works out to
. Evaluation of the phase integrals is more complicated, and the interested reader is directed to the references. The relativistic energy works out to 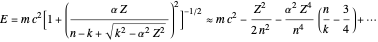 . The third term in the expansion represents the fine-structure splitting, which removes the degeneracy in
. The third term in the expansion represents the fine-structure splitting, which removes the degeneracy in 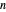 for
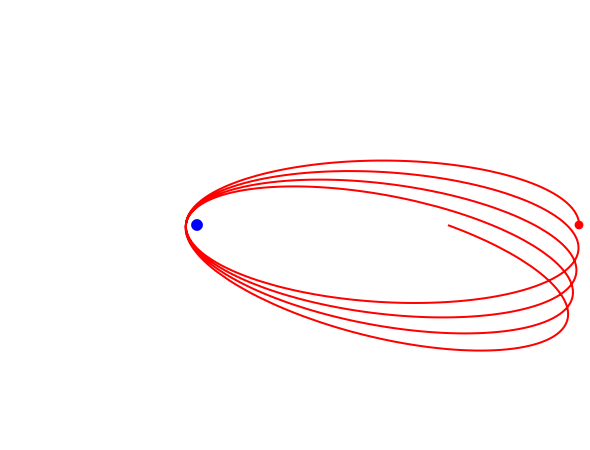
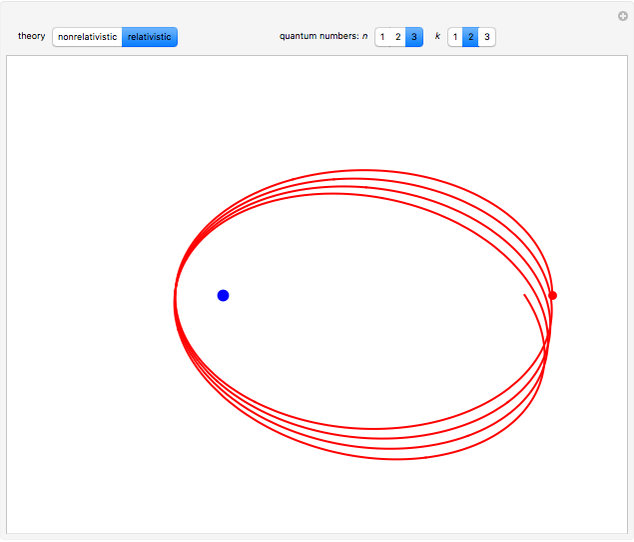
for 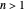 . In the relativistic theory, the elliptical orbits precess about their major axes.
. In the relativistic theory, the elliptical orbits precess about their major axes.
Remarkably, Sommerfeld's formula agrees exactly with the result obtained by solution of the Dirac equation for the hydrogen atom in relativistic quantum mechanics. To be blunt, this is really "dumb luck," since Sommerfeld was, at the time, unaware of electron spin, whereas the Dirac formula actually takes account of the electron's spin-orbit coupling. And, of course, the angular momenta in the old quantum theory are too large by one unit of ℏ.
References
[1] A. Sommerfeld, Atombau und Spektrallinien, 4th ed., Braunschweig, Germany: F. Vieweg & Sohn, 1924 pp. 504–521.
[2] L. Page, Introduction to Theoretical Physics, 3rd ed., Princeton: Van Nostrand, 1952 pp. 670–680.
Permanent Citation