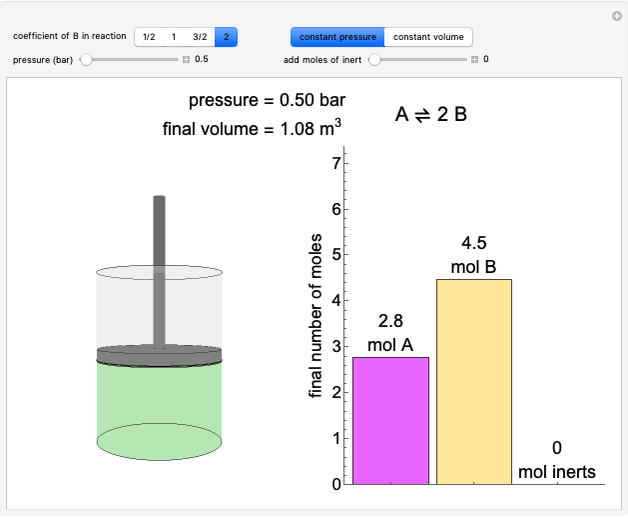
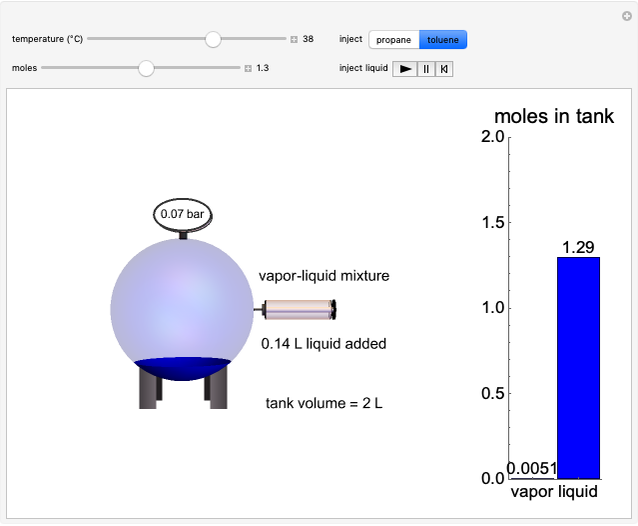
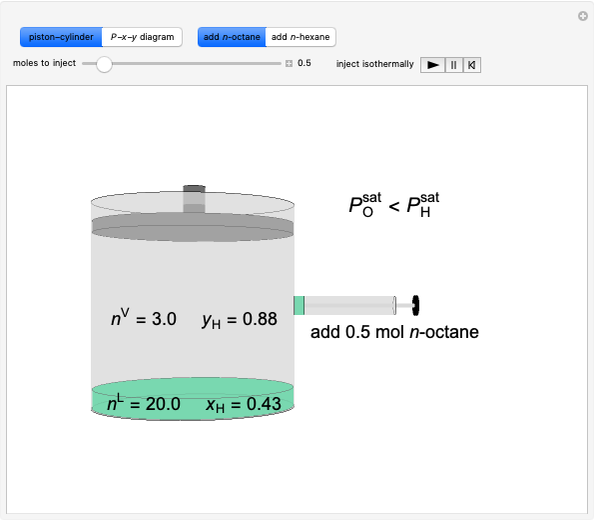
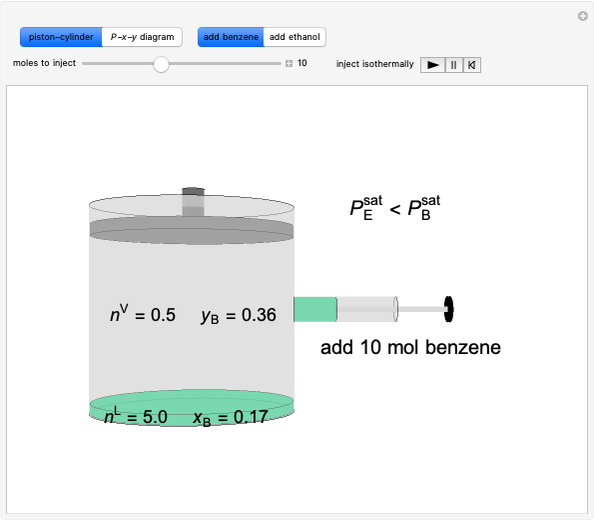
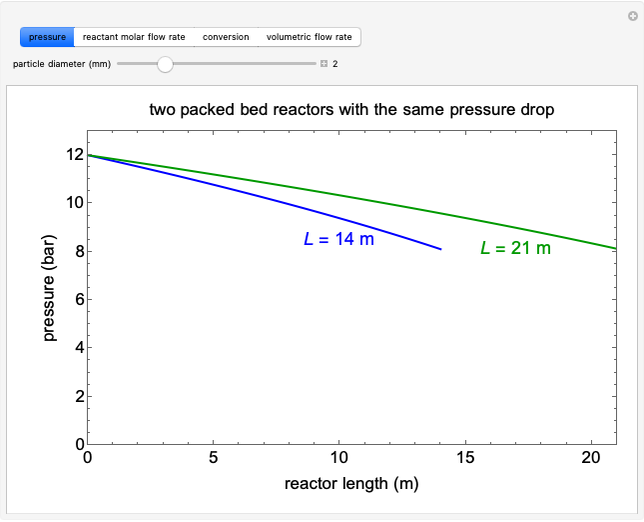
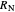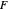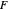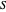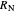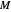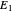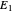Liquid-Liquid Extraction (LLE) on a Right-Triangle Ternary Phase Diagram

Requires a Wolfram Notebook System
Interact on desktop, mobile and cloud with the free Wolfram Player or other Wolfram Language products.
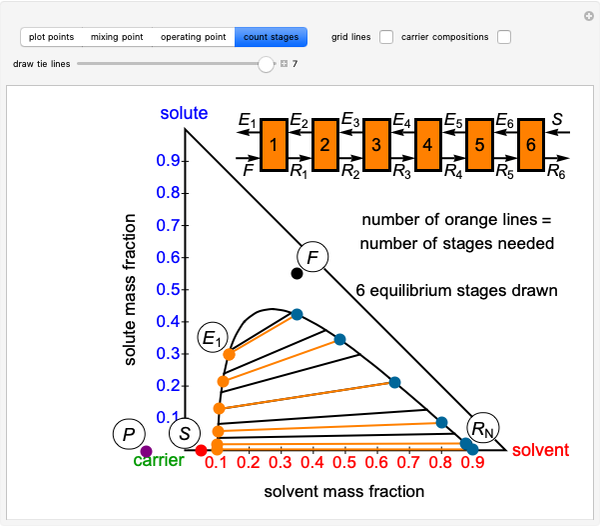
This Demonstration shows how to use the Hunter–Nash method on a right-triangle ternary phase diagram.
[more]
Contributed by: Rachael L. Baumann (September 2017)
Additional contributions by: John L. Falconer
(University of Colorado Boulder, Department of Chemical and Biological Engineering)
Open content licensed under CC BY-NC-SA
Snapshots
Details
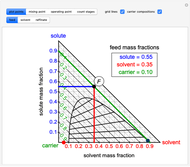
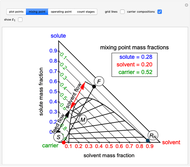
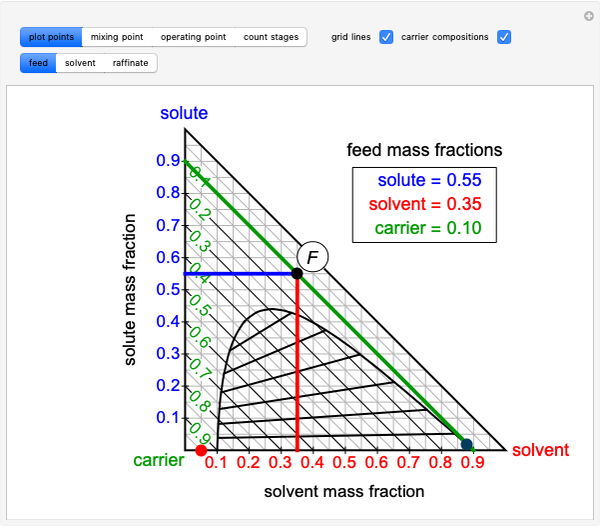
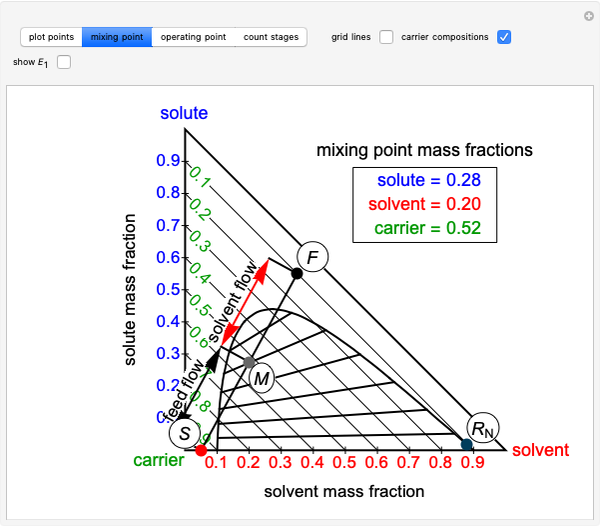
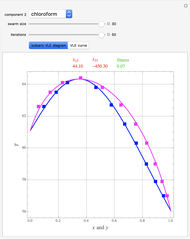
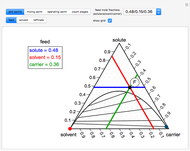
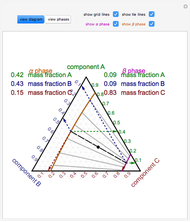
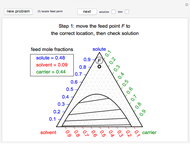
The feed composition is selected within a narrow range of compositions, but the solvent composition and the desired raffinate composition are specified. The extract composition and the number of stages to obtain the desired raffinate composition are determined by mass balances. First, the composition of the mixing point is calculated and located on the ternary phase diagram. The mixing point corresponds to the composition that would be obtained if the feed and the solvent flows were mixed together.
The mixing point 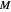 is located on the phase diagram between the feed and solvent points:
is located on the phase diagram between the feed and solvent points:
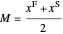 ,
,
where 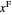 and
and 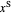 are the feed and solvent compositions.
are the feed and solvent compositions.
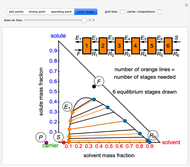
A line is drawn from the desired raffinate composition 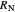 through the mixing point until it intersects the phase boundary; this gives the extract composition
through the mixing point until it intersects the phase boundary; this gives the extract composition 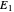 leaving stage 1.
leaving stage 1.
The operating point 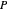 is located at the intersection of a line drawn through points
is located at the intersection of a line drawn through points 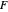 and
and 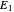 and a line drawn through
and a line drawn through 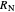 and
and  because the overall mass balance for the system is:
because the overall mass balance for the system is:
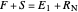 .
.
This equation is rearranged to determine the operating point:
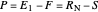 .
.
A tie line from 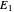 to the right side of the phase boundary yields the raffinate composition
to the right side of the phase boundary yields the raffinate composition 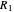 leaving stage 1; this line represents the first equilibrium stage (orange).
leaving stage 1; this line represents the first equilibrium stage (orange).
A mass balance in stage 1 (total feed in = total feed out) is:
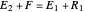 ,
,
which can be written as:
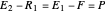 .
.
Thus, the extract composition 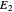 leaving stage 2 is found by drawing a straight line from
leaving stage 2 is found by drawing a straight line from 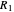 to
to 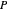 (because
(because 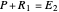 from the previous equation). Where this line intersects the left side of the phase boundary is the composition of
from the previous equation). Where this line intersects the left side of the phase boundary is the composition of 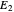 .
.
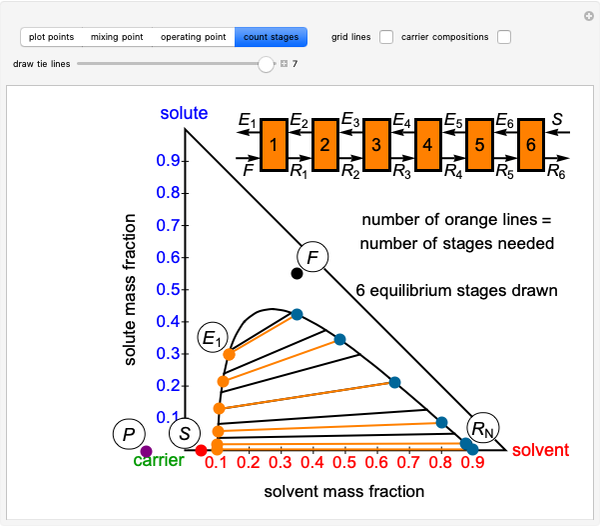
This procedure is repeated for additional stages until the raffinate composition is nearly equal to the desired value 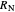 without being less than
without being less than 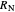 . The number of orange equilibrium lines drawn is the number of equilibrium stages needed to obtain
. The number of orange equilibrium lines drawn is the number of equilibrium stages needed to obtain 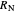 .
.
See [1–4] for screencasts that describe the Hunter–Nash method and present examples.
References
[1] Hunter–Nash Method: Liquid-Liquid Extraction [Video]. Retrieved July 31, 2017, from www.youtube.com/watch?v=e0Yt5RMkyj4.
[2] Hunter–Nash Method: Liquid-Liquid Extraction Example [Video]. Retrieved July 31, 2017, from www.youtube.com/watch?v=FaKIZU3XPR8.
[3] Hunter–Nash Method 1: Mixing and Operating Points [Video]. Retrieved July 31, 2017, from www.youtube.com/watch?v=-yW0jIcH0_E.
[4] Hunter–Nash Method 2: Number of Stages [Video]. Retrieved July 31, 2017, from www.youtube.com/watch?v=JZuavrt8ksQ.
Permanent Citation