DMSO as an Entrainer to Separate a Water-Isopropyl Alcohol Mixture

Requires a Wolfram Notebook System
Interact on desktop, mobile and cloud with the free Wolfram Player or other Wolfram Language products.
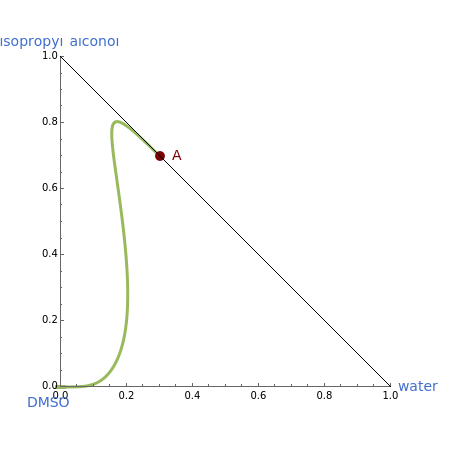
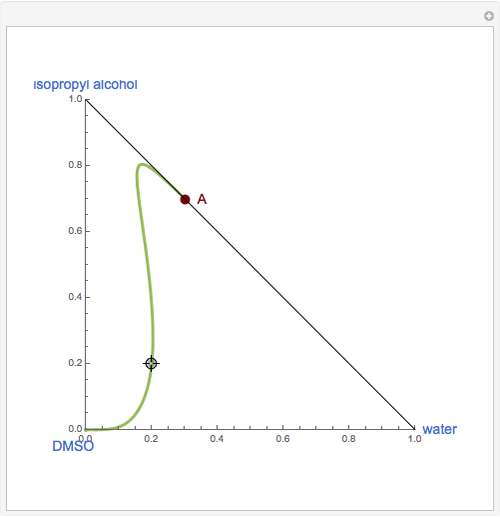
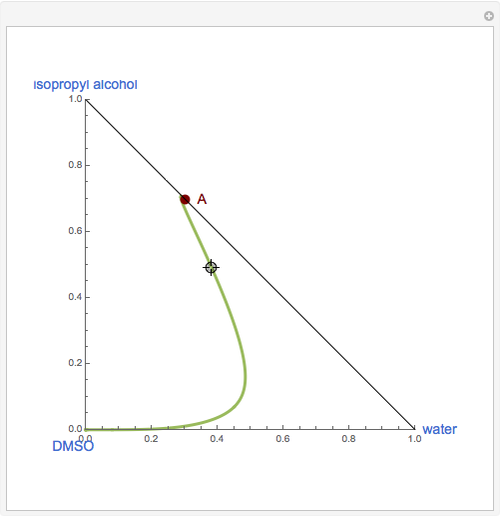
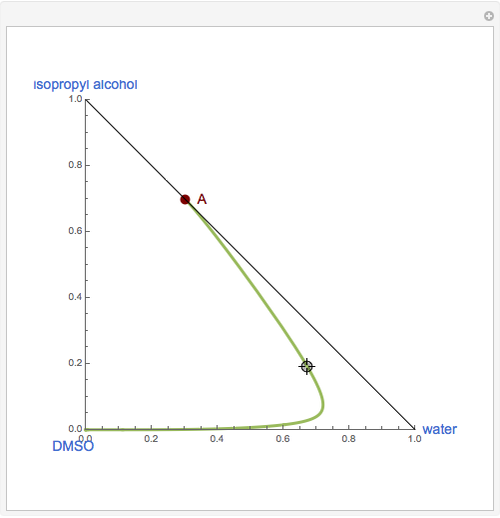
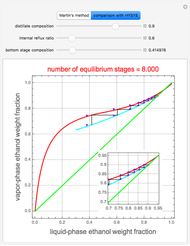
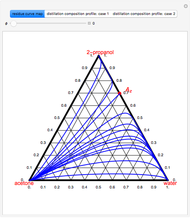
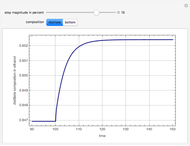
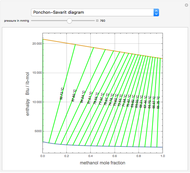
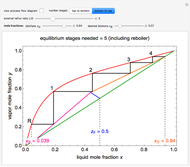
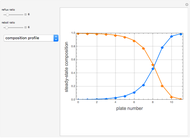
The binary mixture of water and isopropyl alcohol (isopropanol or propan-2-ol) presents a positive azeotrope at 1 atm. This azeotrope is labeled 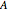 in the ternary diagram and contains
in the ternary diagram and contains 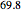 mole % isopropanol. It is possible to break this azeotrope using extractive distillation. A list of entrainers includes: (1) 1,2-ethanediol or ethylene glycol, (2) DSMO (dimethyl sulfoxide), (3)
mole % isopropanol. It is possible to break this azeotrope using extractive distillation. A list of entrainers includes: (1) 1,2-ethanediol or ethylene glycol, (2) DSMO (dimethyl sulfoxide), (3) 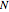 -methyl-2-pyrrolidone or NMP, (4)
-methyl-2-pyrrolidone or NMP, (4) 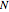 -methyl-2-piperidone, and (5)
-methyl-2-piperidone, and (5) 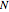 -methyl-6-caprolactam; see [1].
-methyl-6-caprolactam; see [1].
Contributed by: Housam Binous (March 2011)
(Kind Fahd University Petroleum & Minerals)
Open content licensed under CC BY-NC-SA
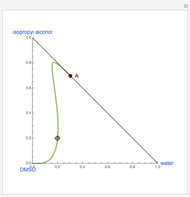
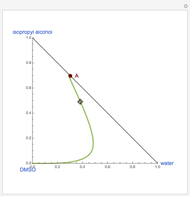
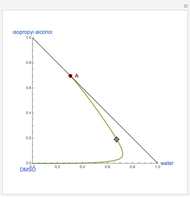
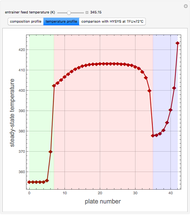
Snapshots
Details
Reference
[1] J. Gmehling and C. Mollmann, "Synthesis of Distillation Process Using Thermodynamic Models and the Dortmund Data Bank," Industrial and Engineering Chemistry Research, 37(8), 1998 pp. 3112–3123.
Permanent Citation