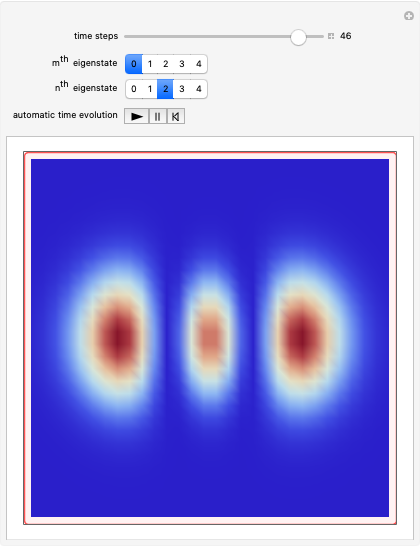
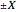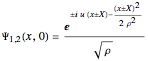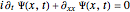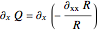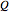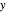Probably one of the most interesting and fundamental wave-particle duality problems in modern physics is the double-slit experiment.
In this Demonstration the double‐slit experiment is simulated numerically according to the causal interpretation of David Bohm. The slits generate two Gaussian profiles in  space, positioned at
space, positioned at 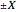 . The initial form of the unnormalized waves is
. The initial form of the unnormalized waves is 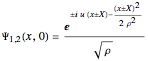 , where
, where  is the velocity in the
is the velocity in the  direction and
direction and  is the width of the wave packet at
is the width of the wave packet at  . In this case the total wavefunction is the superposition of the two waves, where the time evolution is to be calculated from the free Schrödinger equation:
. In this case the total wavefunction is the superposition of the two waves, where the time evolution is to be calculated from the free Schrödinger equation: 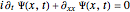 .
.
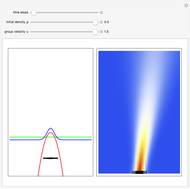
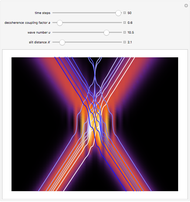
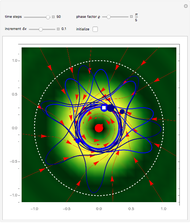
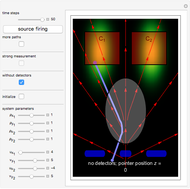
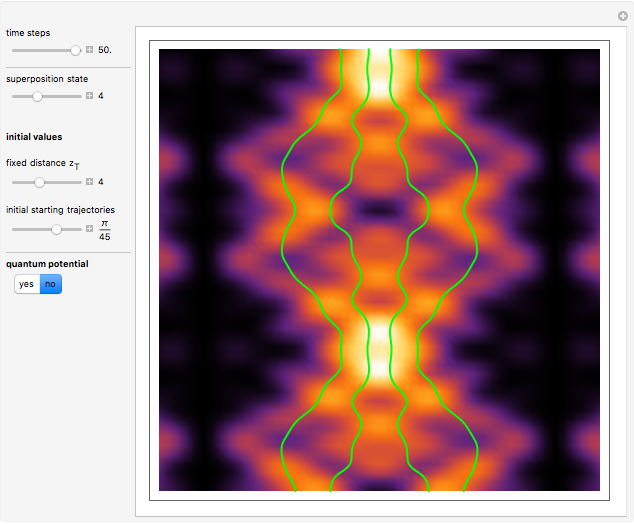
According to the orthodox interpretation of quantum theory, it cannot be decided which slit the particle goes through because of the self-interference effect. It has to be assumed that the particle goes through both slits and the wavefunction collapses when a measurement is made at the detector screen.
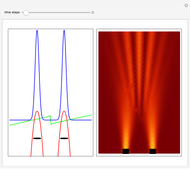
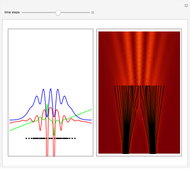
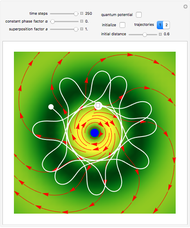
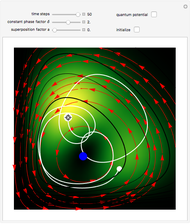
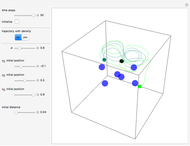
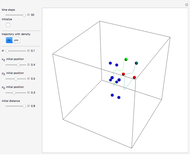
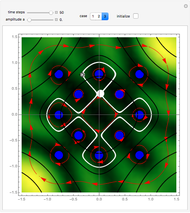
The Bohmian interpretation is completely different. The quantum particle passes through slit one or slit two and never crosses the axis of symmetry. The paths of the quantum particles are not detectable directly because of the perturbation effects of the measuring process. Which slit the particles pass through depends on the initial position only. The quantum particles possess well-defined positions and velocities at all times. In the causal interpretation of quantum theory there is a quantum force proportional to 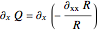 , where
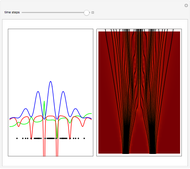
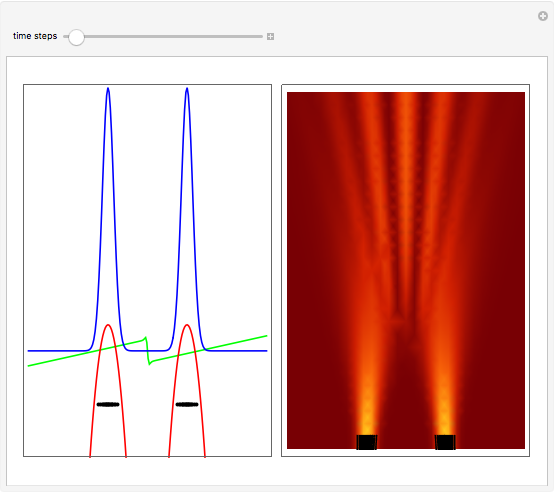
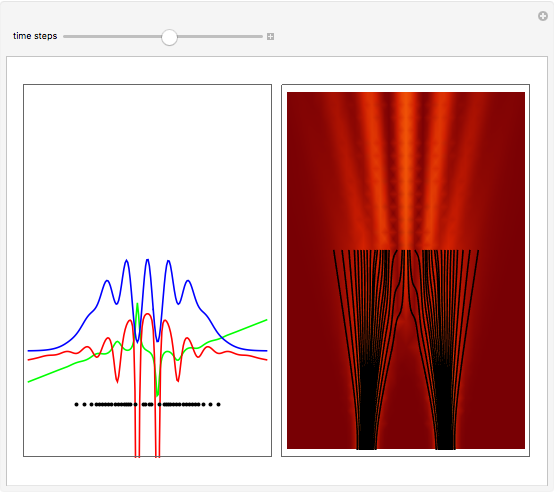
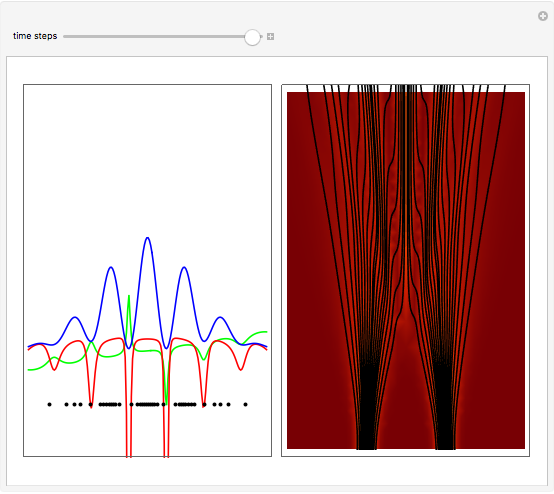
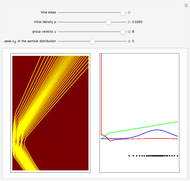
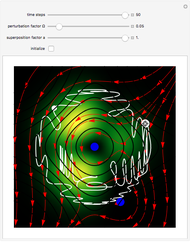
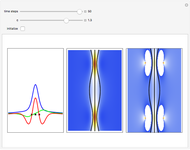
, where 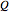 is called the quantum potential. The quantum potential leads to highly nonclassical motion of particles. The trajectories run to the local maxima of the squared wavefunction and therefore correspond to the bright fringes of the diffraction pattern.
is called the quantum potential. The quantum potential leads to highly nonclassical motion of particles. The trajectories run to the local maxima of the squared wavefunction and therefore correspond to the bright fringes of the diffraction pattern.
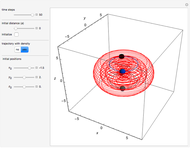
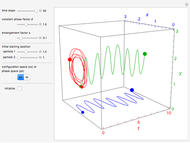
The trajectories in the ( ,
,  ) space do not display the real space, but the structure in the real (
) space do not display the real space, but the structure in the real ( ,
, 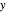 ) space is the same. The motion of the particles is obtained by integrating the gradient of the real phase function from the final wavefunction in the eikonal representation.
) space is the same. The motion of the particles is obtained by integrating the gradient of the real phase function from the final wavefunction in the eikonal representation.
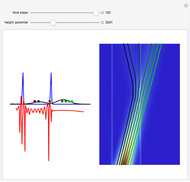
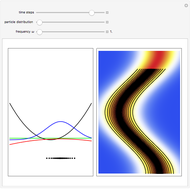
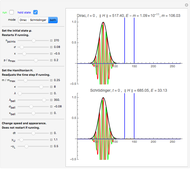
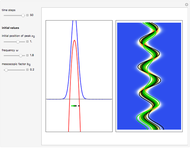
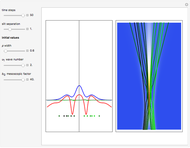
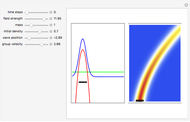
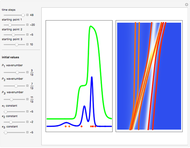
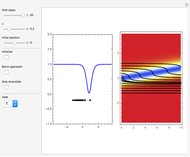
On the right side, the graphic shows the squared wavefunction and the trajectories. The left side shows the particles' position, the squared wavefunction (blue), the quantum potential (red), and the velocity (green).
[less]