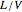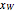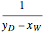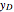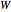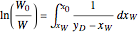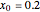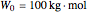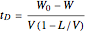Three-Stage Batch Rectification with Constant Reflux
Initializing live version

Requires a Wolfram Notebook System
Interact on desktop, mobile and cloud with the free Wolfram Player or other Wolfram Language products.
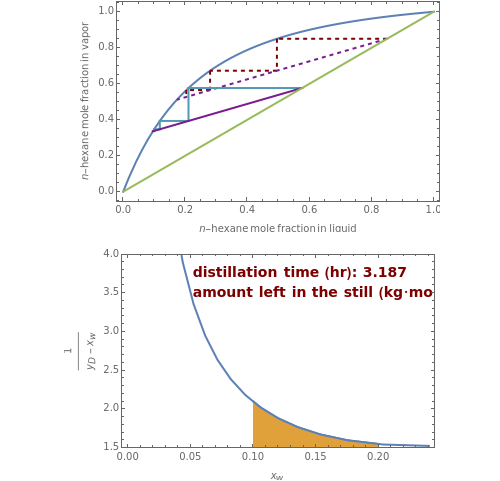
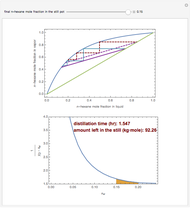
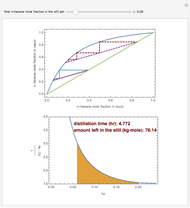
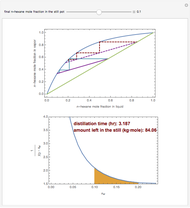
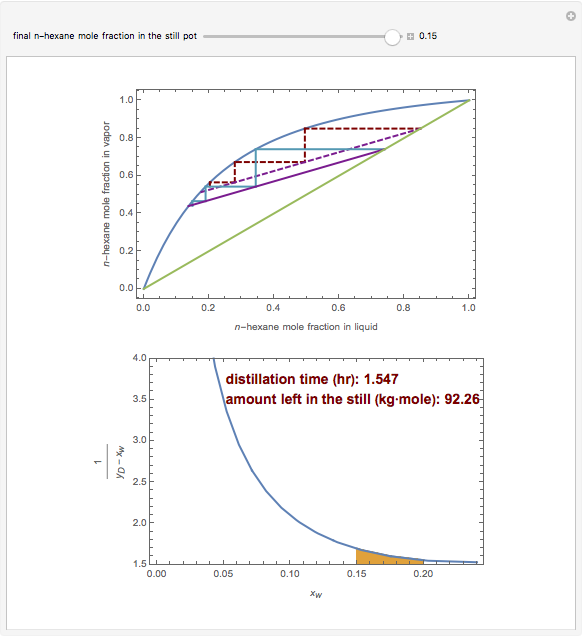
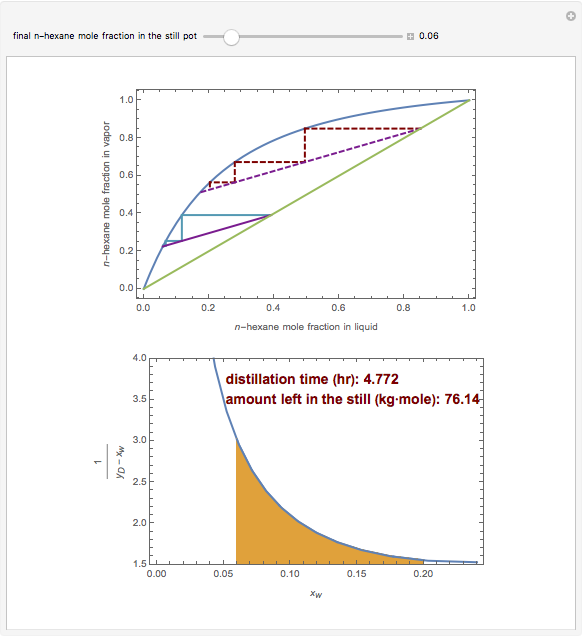
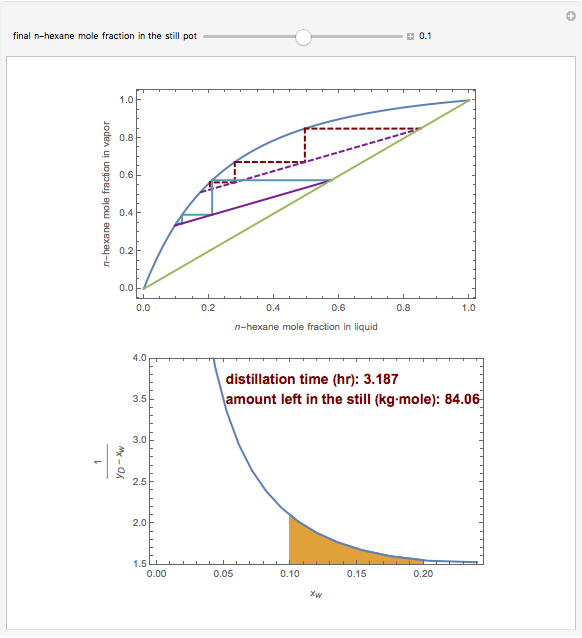
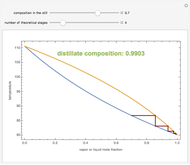
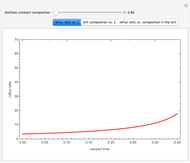
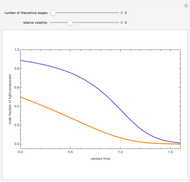
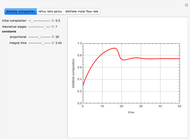
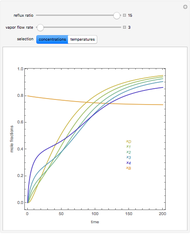
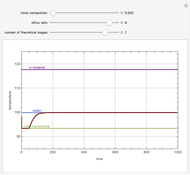
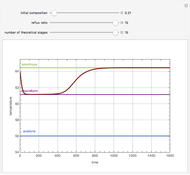
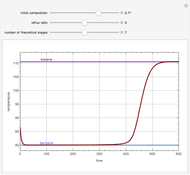
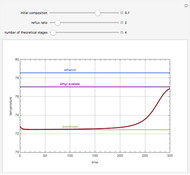
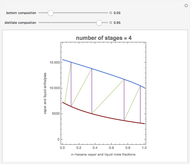
Consider a three-stage batch rectification column charged with 100 kg·mole of a binary mixture composed of 20% mole fraction of  -hexane and 80% mole fraction of
-hexane and 80% mole fraction of  -octane. The boilup rate,
-octane. The boilup rate, 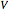 , is constant and is set equal to 10 kg·mole/hr. The constant reflux ratio is equal to 1.
, is constant and is set equal to 10 kg·mole/hr. The constant reflux ratio is equal to 1.
Contributed by: Housam Binous (March 2011)
Open content licensed under CC BY-NC-SA
Snapshots
Details
E. J. Henley and J. D. Seader, Equilibrium-Stage Separations in Chemical Engineering, New York: John Wiley & Sons, 1981.
Permanent Citation