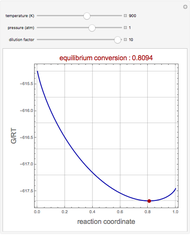
Construct a Pressure-Composition Diagram for Immiscible Liquids

Requires a Wolfram Notebook System
Interact on desktop, mobile and cloud with the free Wolfram Player or other Wolfram Language products.
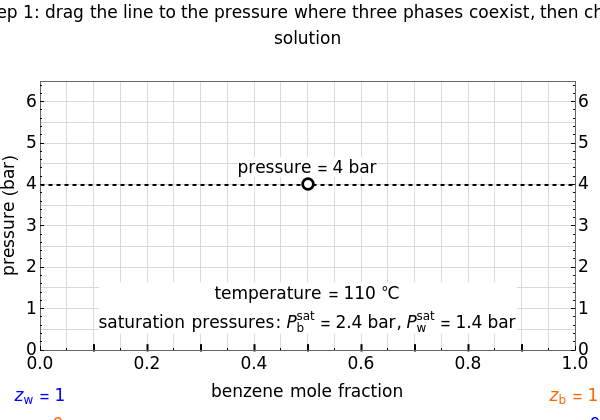
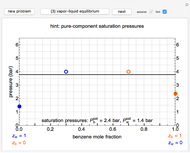
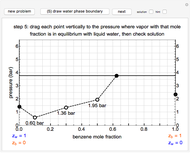
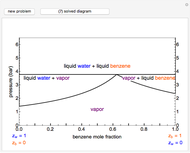
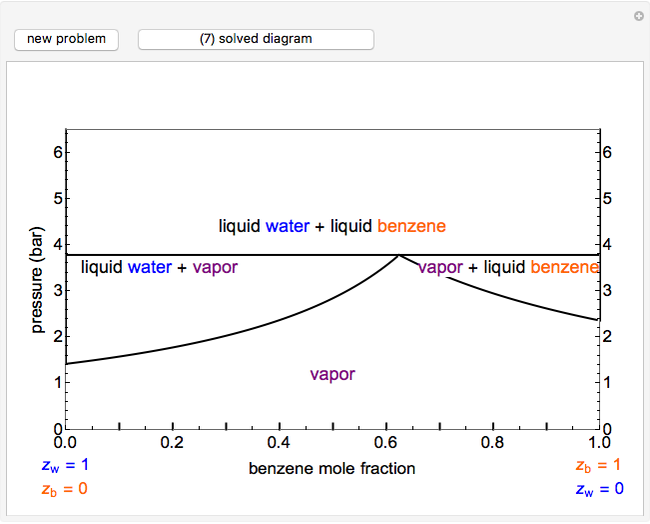
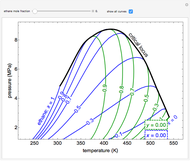
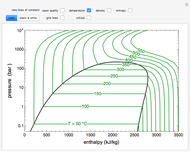
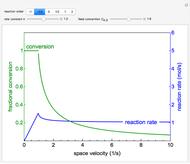
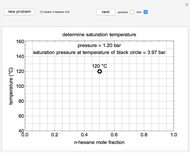
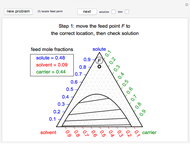
This Demonstration leads you through a step-by-step procedure to create a pressure-composition diagram for two immiscible liquids (water and an organic compound) at a fixed temperature. The organic can be benzene, toluene or  -hexane. In each step, drag a line or point to make a guess and then check the "solution" box to show the correct answer. You can only move forward or select "new problem" to start over at a different temperature and a different organic. For any step, check "hint" for help.
-hexane. In each step, drag a line or point to make a guess and then check the "solution" box to show the correct answer. You can only move forward or select "new problem" to start over at a different temperature and a different organic. For any step, check "hint" for help.
Contributed by: Rachael L. Baumann (March 2018)
Additional contributions by: John L. Falconer
(University of Colorado Boulder, Department of Chemical and Biological Engineering)
Open content licensed under CC BY-NC-SA
Snapshots
Details
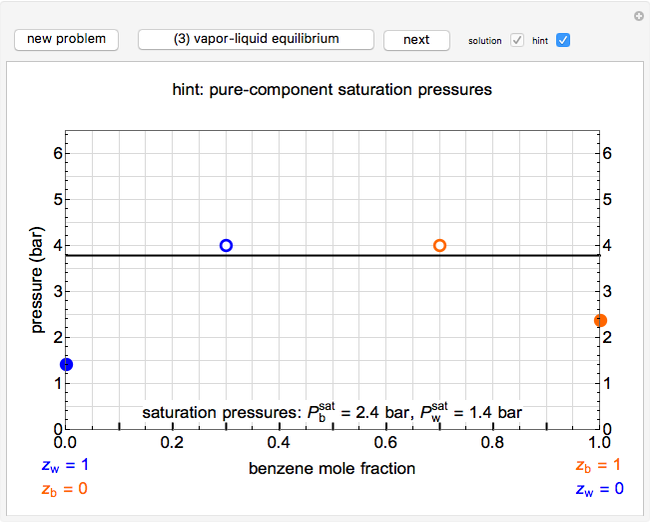
Immiscible components do not mix in the liquid phase, and each independently exerts its saturation pressure  and
and 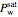 ; the subscripts
; the subscripts 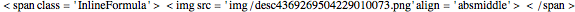 and
and 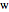 refer to benzene and water. The equations are the same for the other organics (toluene,
refer to benzene and water. The equations are the same for the other organics (toluene,  -hexane). The total pressure
-hexane). The total pressure 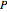 above the two immiscible liquids is equal to the sum of their saturation pressures:
above the two immiscible liquids is equal to the sum of their saturation pressures:
 .
.
The saturation pressures are calculated using the Antoine equation:
 ,
,
where  is the saturation pressure of
is the saturation pressure of 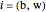 (bar);
(bar);  ,
, 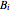 and
and 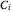 are Antoine constants; and
are Antoine constants; and  is temperature (°C).
is temperature (°C).
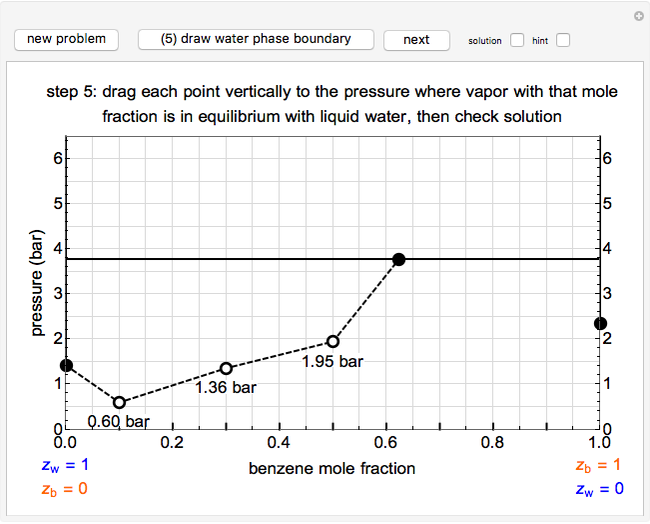
For the benzene-water system, for conditions where benzene condenses, the dew point curve is:
 ,
,
where  is the mole fraction of benzene in the vapor phase.
is the mole fraction of benzene in the vapor phase.
For conditions where water condenses, the dew point curve is:
 ,
,
where  is the mole fraction of water in the vapor phase and
is the mole fraction of water in the vapor phase and 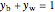 .
.
The screencast video at [1] explains how to use a similar Demonstration.
Reference
[1] Immiscible Liquids on Pressure-Composition Diagram [Video]. (Feb 1, 2018) www.colorado.edu/learncheme/thermodynamics/ImmiscibleLiquidsPxy.html.
Permanent Citation