Fugacity from Equation of State for Water

Requires a Wolfram Notebook System
Interact on desktop, mobile and cloud with the free Wolfram Player or other Wolfram Language products.
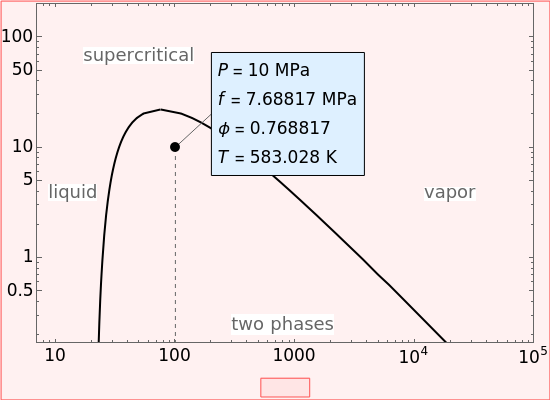
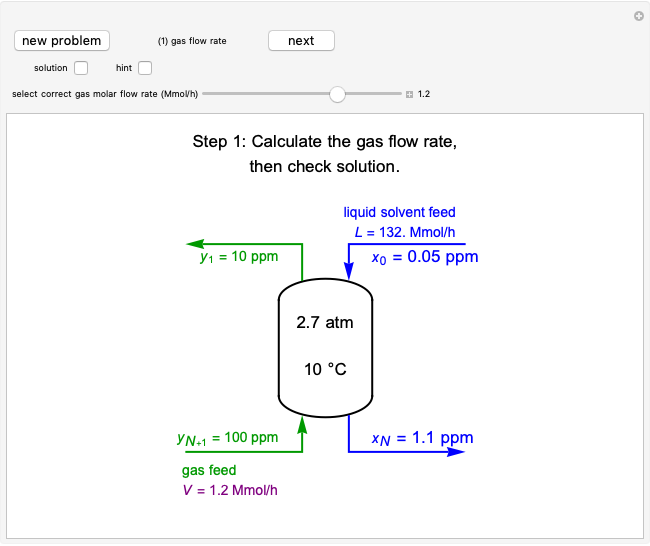
The fugacity  and the fugacity coefficient
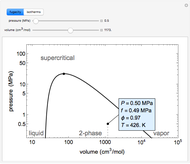
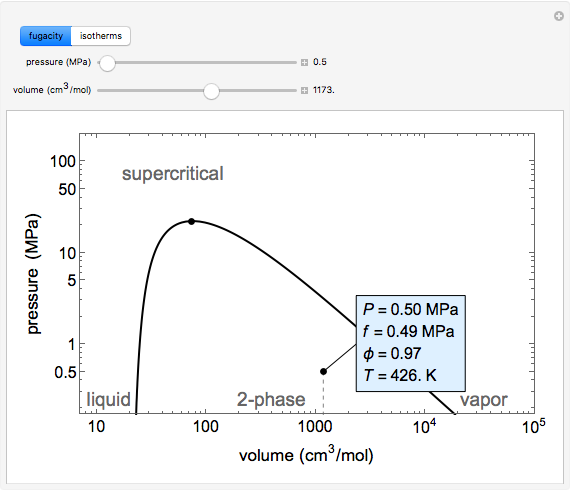
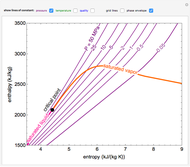
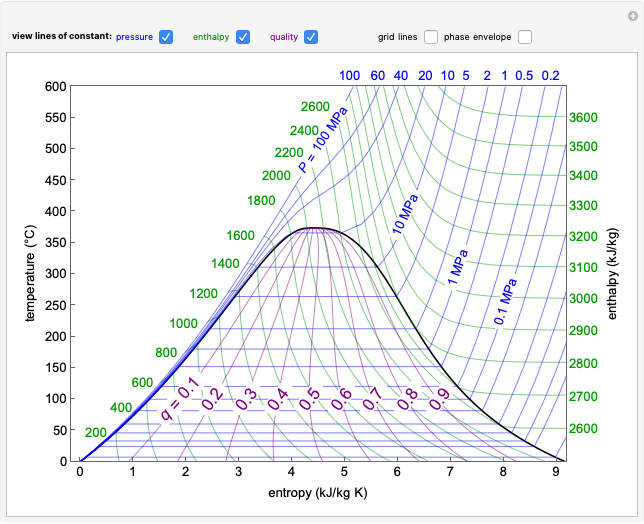
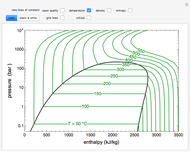
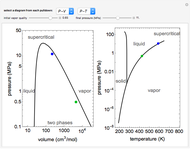
and the fugacity coefficient  are calculated for water using the Peng–Robinson equation of state (EOS). Select "fugacity" and change pressure and volume with sliders; the corresponding state is indicated by the black dot on the log pressure versus log volume diagram. Change the volume more slowly by holding down the ALT key while moving the slider. The fugacity coefficient indicates how much the fluid deviates from ideal-gas behavior (
are calculated for water using the Peng–Robinson equation of state (EOS). Select "fugacity" and change pressure and volume with sliders; the corresponding state is indicated by the black dot on the log pressure versus log volume diagram. Change the volume more slowly by holding down the ALT key while moving the slider. The fugacity coefficient indicates how much the fluid deviates from ideal-gas behavior ( for an ideal gas). Select "isotherms" to display isotherms on the
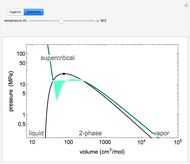
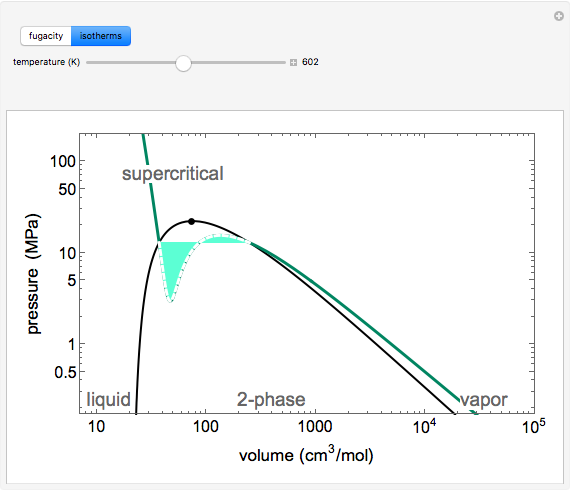
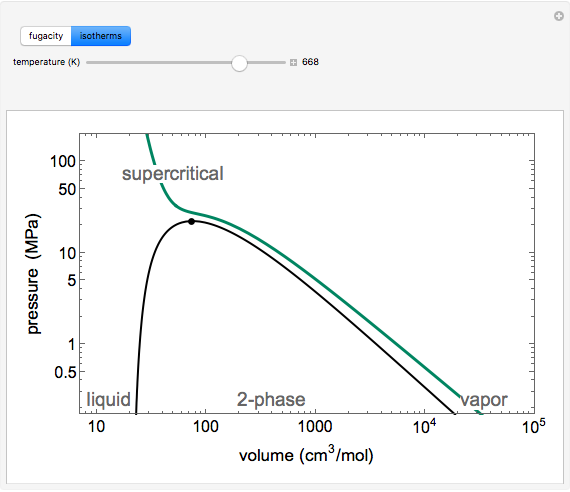
for an ideal gas). Select "isotherms" to display isotherms on the  diagram and change temperature with a slider; the horizontal blue line represents liquid and vapor in equilibrium. The yellow areas between the isotherm and the horizontal blue line are equal when viewed on a linear scale.
diagram and change temperature with a slider; the horizontal blue line represents liquid and vapor in equilibrium. The yellow areas between the isotherm and the horizontal blue line are equal when viewed on a linear scale.
Contributed by: Rachael L. Baumann (January 2015)
With additional contributions by: John L. Falconer and Nick Bongiardina
(University of Colorado Boulder, Department of Chemical and Biological Engineering)
Open content licensed under CC BY-NC-SA
Snapshots
Details
The Peng–Robinson EOS is used to calculate pressure:
 ,
,
where  is pressure (MPa),
is pressure (MPa),  is the ideal gas constant (
is the ideal gas constant ( ),
),  is temperature (K),
is temperature (K),  is volume (
is volume ( ) and
) and  and
and  are constants:
are constants:
 ,
,
 ,
,
 .
.
Here  is a constant associated with the acentric factor
is a constant associated with the acentric factor  ,
,  is the critical temperature (K) and
is the critical temperature (K) and  is the critical pressure (MPa).
is the critical pressure (MPa).
The following equation is solved in order to obtain the compressibility factor 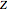 :
:
 ,
,
where  ,
,  ,
,  ,
,  and
and  are all constants:
are all constants:
 ,
,
 ,
,
 ,
,
 ,
,
 .
.
The fugacity  and activity coefficient
and activity coefficient  are calculated by:
are calculated by:
 ,
,
 .
.
When the EOS has three roots, the correct solution is the one that has the lowest fugacity.
A screencast video [1] shows how to use this Demonstration.
Reference
[1] Fugacity from Equation of State for Water [Video]. (Sep 23, 2016) www.colorado.edu/learncheme/thermodynamics/FugacityEOSWater.html.
Permanent Citation