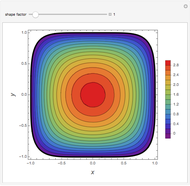
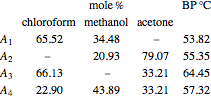Locus of Binary and Ternary Azeotropes in Chloroform-Methanol-Acetone Mixture
Initializing live version

Requires a Wolfram Notebook System
Interact on desktop, mobile and cloud with the free Wolfram Player or other Wolfram Language products.
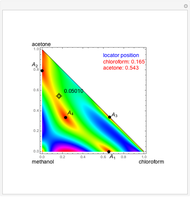
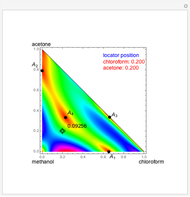
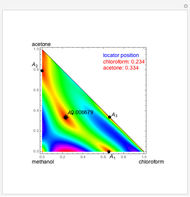
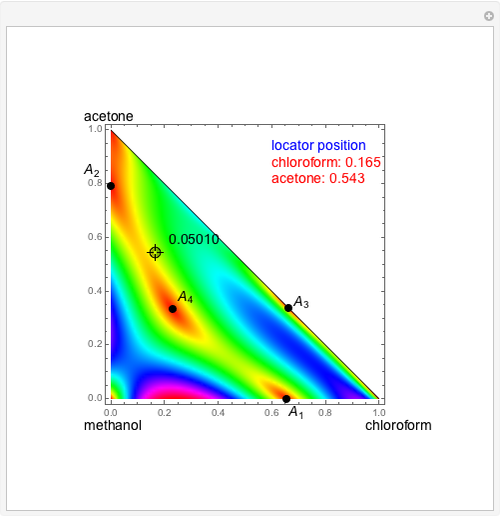
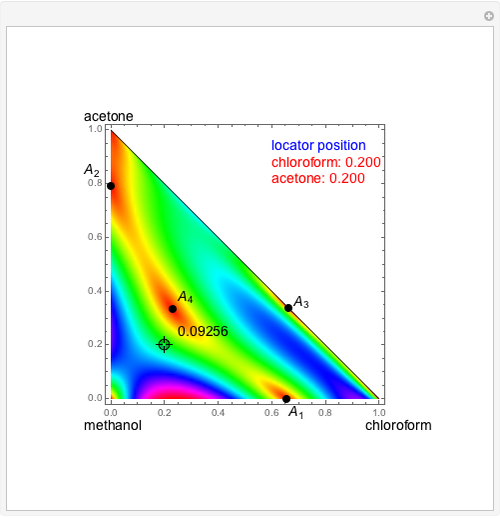
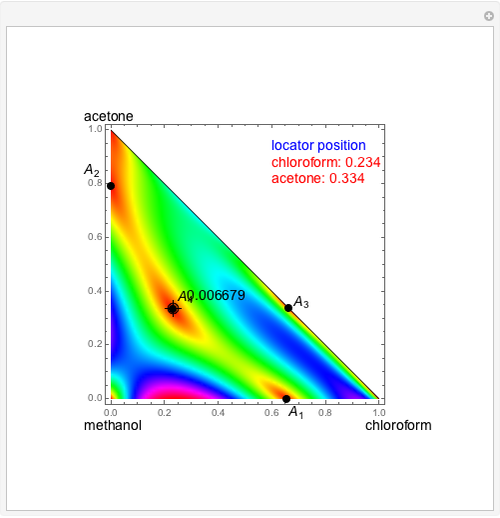
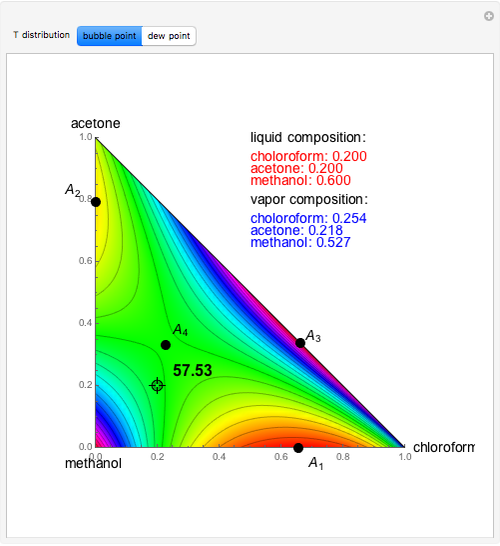
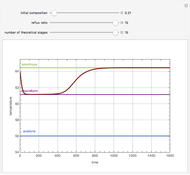
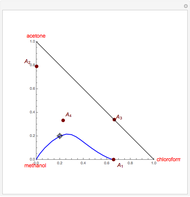
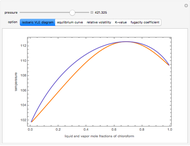
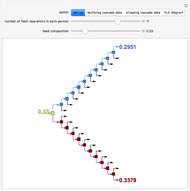
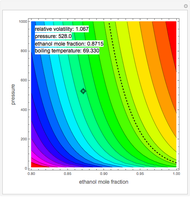
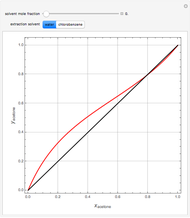
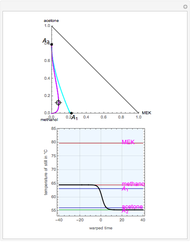
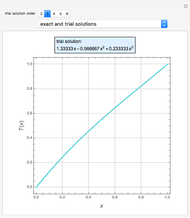
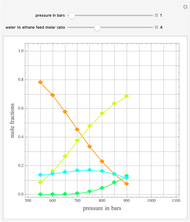
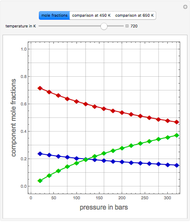
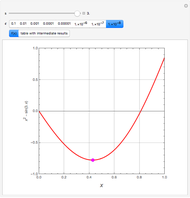
Consider a ternary mixture of chloroform, acetone, and methanol at 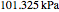 . The vapor-liquid equilibrium (VLE) behavior is described by a modified form of Raoult's law with activity coefficients predicted by the Wilson model [1].
. The vapor-liquid equilibrium (VLE) behavior is described by a modified form of Raoult's law with activity coefficients predicted by the Wilson model [1].
Contributed by: Housam Binous and Brian G. Higgins (September 2011)
Open content licensed under CC BY-NC-SA
Snapshots
Details
Reference:
[1] G. M. Wilson, "Vapor-Liquid Equilibrium XI: A New Expression for the Excess Free Energy of Mixing," Journal of the American Chemical Society, 86(2), 1964 pp. 127–130.
Permanent Citation