Maximizing Conversion to an Intermediate Product in a Two-Stage CSTR

Requires a Wolfram Notebook System
Interact on desktop, mobile and cloud with the free Wolfram Player or other Wolfram Language products.
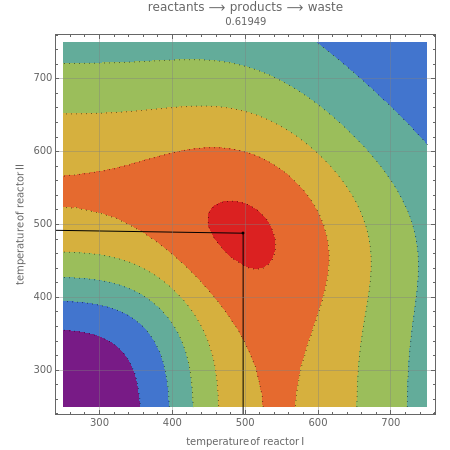
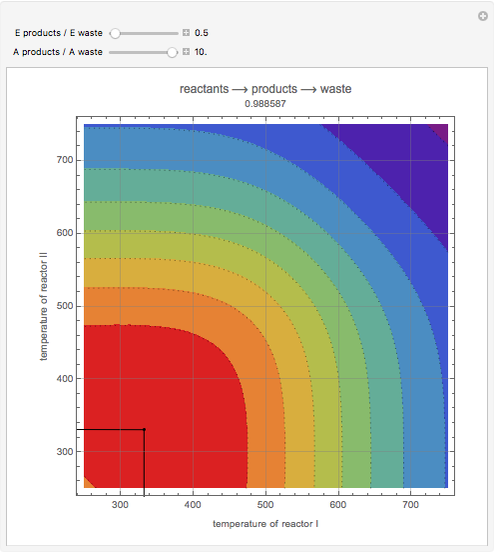
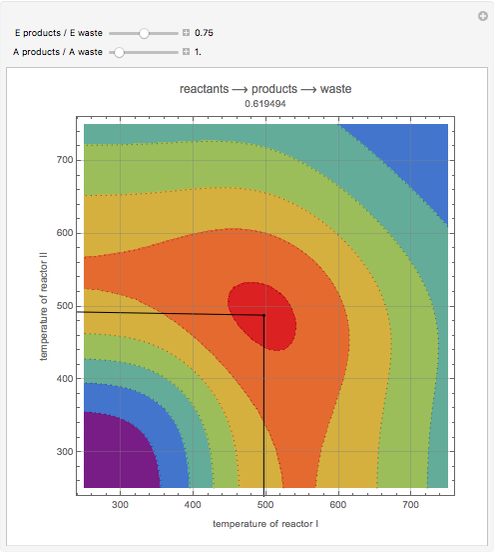
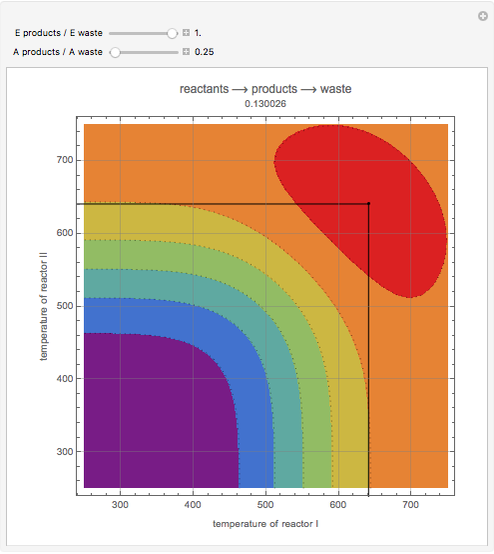
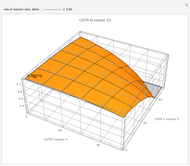
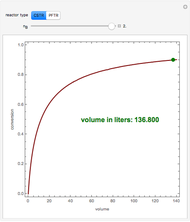
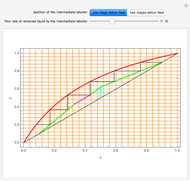
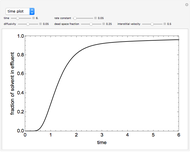
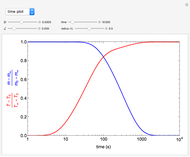
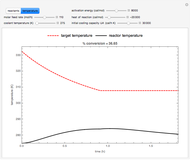
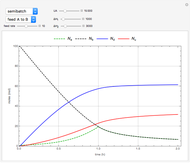
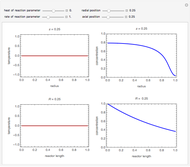
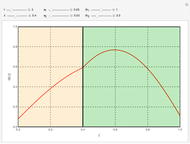
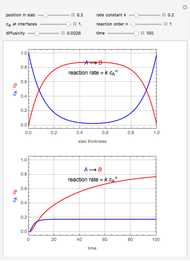
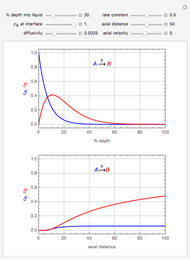
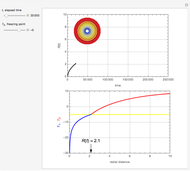
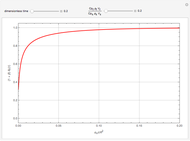
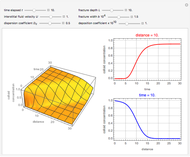
This Demonstration examines a two-stage continuous-flow stirred tank reactor (CSTR). Conversion to an intermediate product of consecutive chemical reactions can be maximized in a combination of two CSTRs operating in series.
[more]
Contributed by: Clay Gruesbeck (August 2012)
Open content licensed under CC BY-NC-SA
Snapshots
Details
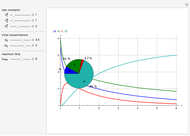
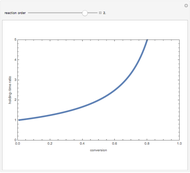
Consider the consecutive first-order chemical reaction  with
with  ,
,  , and
, and  representing reactants, products, and waste, involving the equations
representing reactants, products, and waste, involving the equations  and
and  . Here
. Here  ,
,  , and
, and  are the rate constants, Arrhenius constants, and activation energies of the products and reactants, and
are the rate constants, Arrhenius constants, and activation energies of the products and reactants, and  is the temperature.
is the temperature.
The equations describing the steady state condition of the CSTR are:
 and
and  ,
where
,
where  and
and  are the concentrations,
are the concentrations,  is the flow rate, and
is the flow rate, and  is the reactor volume for reactors
is the reactor volume for reactors  and 2, with initial conditions
and 2, with initial conditions  and
and  .
.
Reference
[1] Wikipedia. "Continuous Reactor." (Aug 21, 2012) en.wikipedia.org/wiki/Continuous_reactor.
Permanent Citation