The Photoelectric Effect

Requires a Wolfram Notebook System
Interact on desktop, mobile and cloud with the free Wolfram Player or other Wolfram Language products.
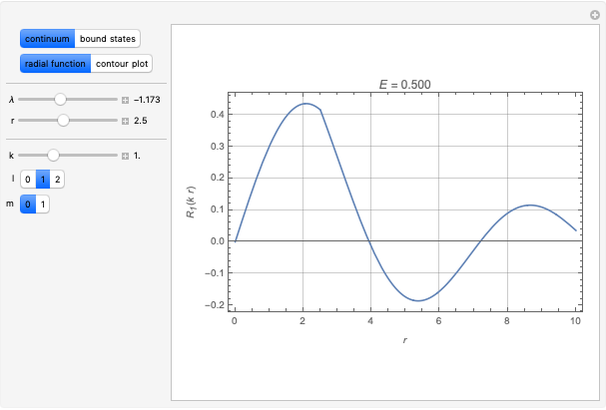
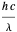 The photoelectric effect is a quantum-mechanical phenomenon in which light impinging on the surface of a metal can cause electrons to be ejected. Only light with wavelengths shorter than some threshold value
The photoelectric effect is a quantum-mechanical phenomenon in which light impinging on the surface of a metal can cause electrons to be ejected. Only light with wavelengths shorter than some threshold value 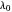 , characteristic of each metal, can cause emission of photoelectrons, no matter how intense the radiation. Einstein explained this in 1905 by proposing that light is composed of discrete photons, each carrying energy
, characteristic of each metal, can cause emission of photoelectrons, no matter how intense the radiation. Einstein explained this in 1905 by proposing that light is composed of discrete photons, each carrying energy 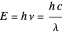 . Only when the photon energy exceeds the work function
. Only when the photon energy exceeds the work function 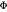 of the metal, a measure of how strongly the outermost electrons are bound, can photoelectrons be emitted. The relevant equation is
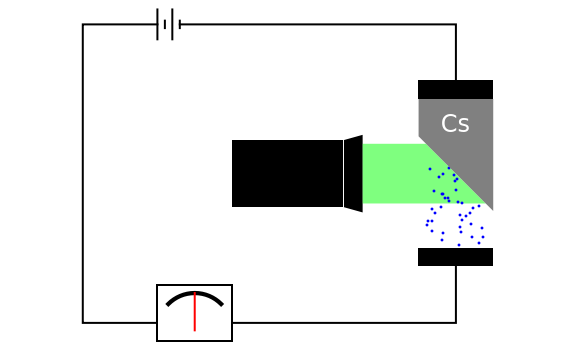
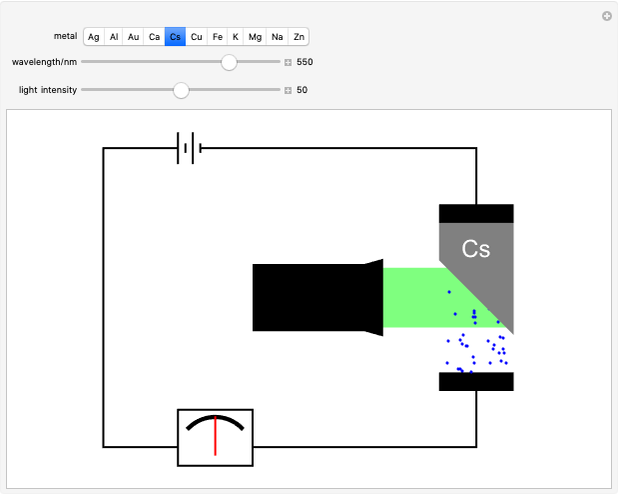
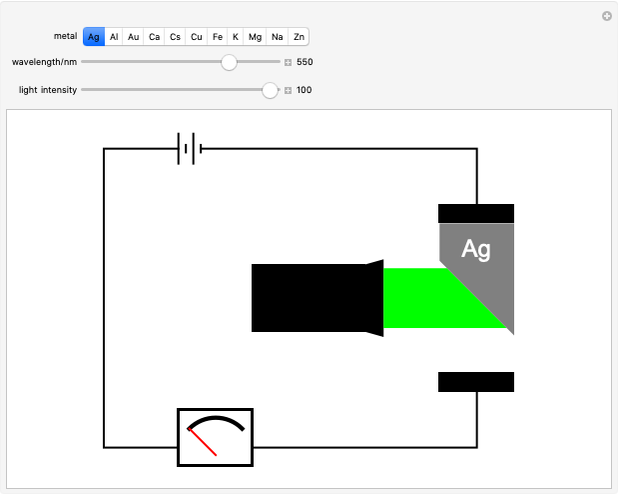
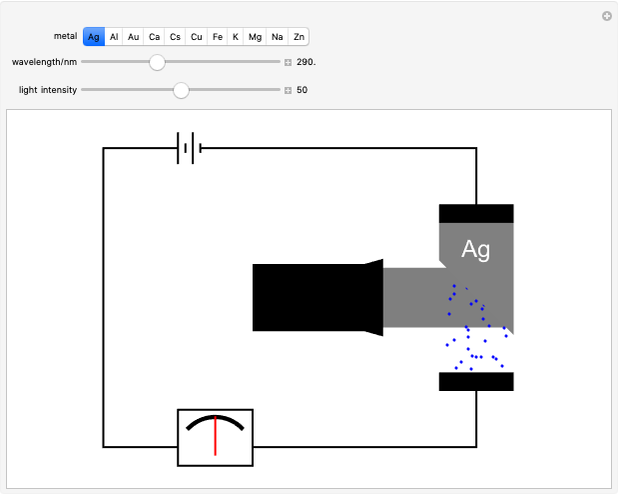
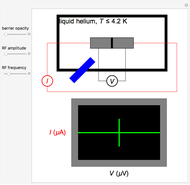
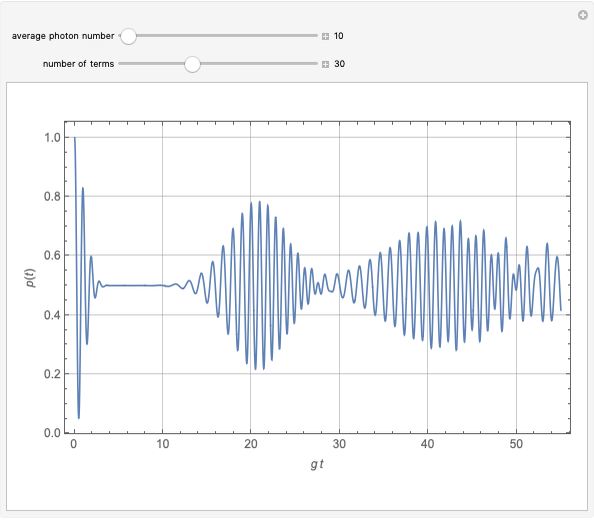
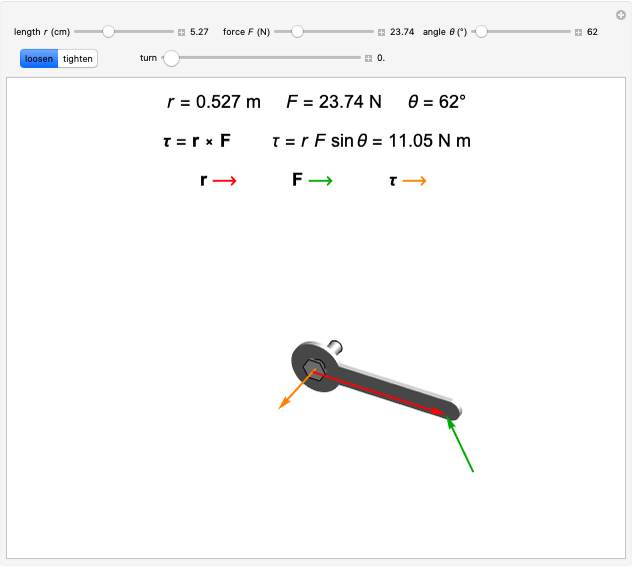
of the metal, a measure of how strongly the outermost electrons are bound, can photoelectrons be emitted. The relevant equation is 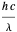 InlineMath=Φ+KEmax, where the last term represents the maximum kinetic energy of the ejected electrons. Once the threshold wavelength is attained, the current of electrons increases linearly with the radiation intensity. This can be monitored by an ammeter in the circuit shown. The light source covers the entire visible range 400–700 nm. In the ultraviolet region, the light ray appears as black.
InlineMath=Φ+KEmax, where the last term represents the maximum kinetic energy of the ejected electrons. Once the threshold wavelength is attained, the current of electrons increases linearly with the radiation intensity. This can be monitored by an ammeter in the circuit shown. The light source covers the entire visible range 400–700 nm. In the ultraviolet region, the light ray appears as black.
Contributed by: S. M. Blinder (March 2008)
Open content licensed under CC BY-NC-SA
Snapshots
Details
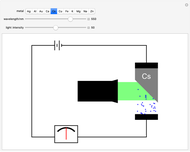
Snapshot 1: above the threshold wavelength, there is no emission of photoelectrons, no matter how high the radiation intensity
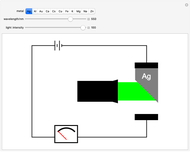
Snapshot 2: at the threshold wavelength for Ag, around 290 nm, the photoelectric effect occurs
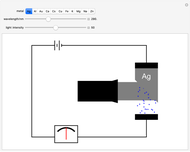
Snapshot 3: once the threshold is reached, the electron current increases linearly with radiation intensity
Permanent Citation