Lemon Batteries

Requires a Wolfram Notebook System
Interact on desktop, mobile and cloud with the free Wolfram Player or other Wolfram Language products.
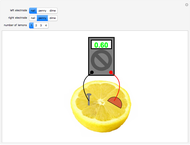
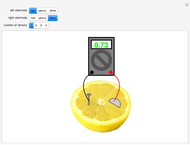
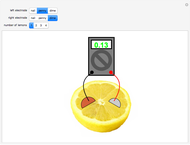
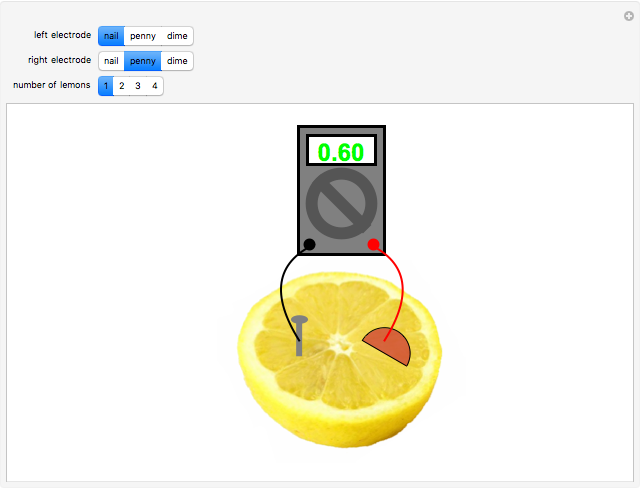
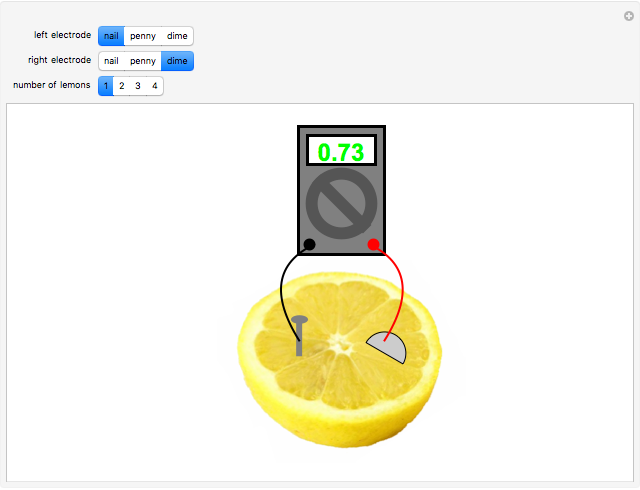
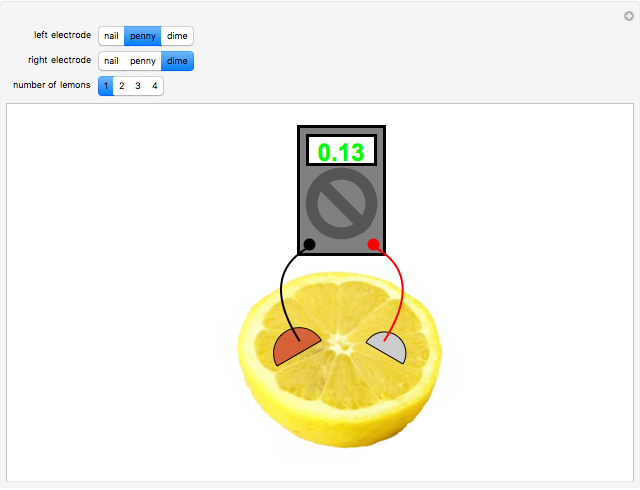
A rudimentary electrochemical cell can be constructed from two dissimilar metals immersed in an electrolyte. The lemon battery is a popular demonstrative example, with the highly acidic juicy insides providing an excellent electrolyte. The potential difference, usually of the order of a fraction of a volt, can be measured by a voltmeter connected to the two electrodes. The values shown in this Demonstration were determined by experiments in the author's laboratory. Combinations of three electrodes were tested: galvanized nails, pennies, and dimes, with compositions approximating those of zinc, copper, and silver, respectively. A positive voltage will be obtained if the left-hand electrode is the more electropositive metal. Reversing the electrodes (or voltmeter connections) gives the negative of the original reading. Two or more lemons can be connected in series to give multicell batteries.
Contributed by: S. M. Blinder (March 2011)
Open content licensed under CC BY-NC-SA
Snapshots
Details
Snapshots: the combinations Zn/Cu, Zn/Ag, Cu/Ag give readings of 0.60, 0.73 and 0.13 V, respectively; if the corresponding pure metals under standard conditions were used, the values should be 1.10, 1.56 and 0.46 V
Permanent Citation
"Lemon Batteries"
http://demonstrations.wolfram.com/LemonBatteries/
Wolfram Demonstrations Project
Published: March 7 2011