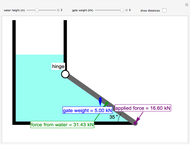
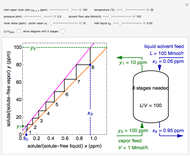
Why Density Change Cannot Be Ignored in a Plug Flow Reactor (PFR)

Requires a Wolfram Notebook System
Interact on desktop, mobile and cloud with the free Wolfram Player or other Wolfram Language products.
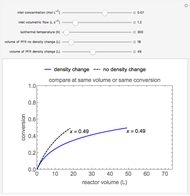
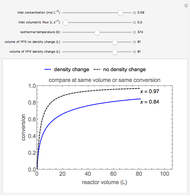
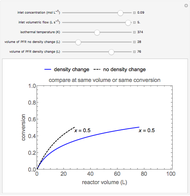
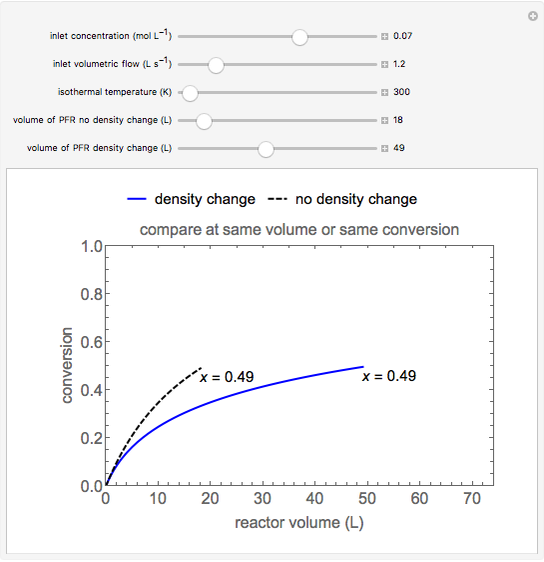
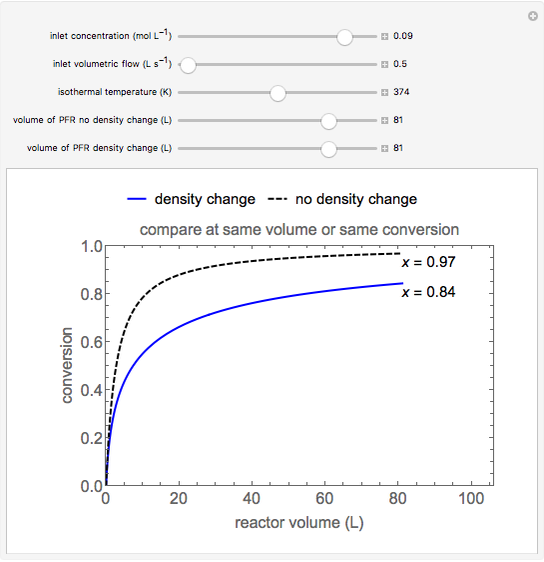
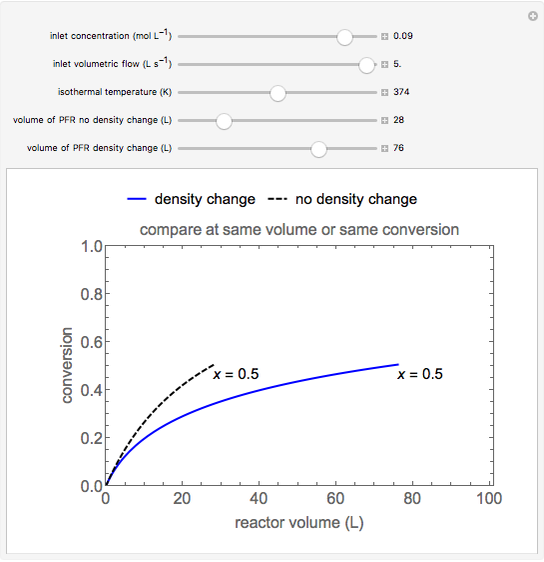
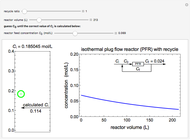
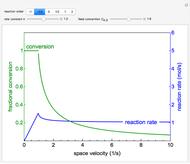
This Demonstration calculates conversion as a function of reactor volume for a second-order, gas-phase reaction in an isothermal, isobaric, plug flow reactor (PFR). The number of moles increases as conversion increases for the reaction 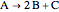 , and this causes the mass density to decrease. Thus, the volumetric flow rate increases down the reactor so that the conversion (blue line) is lower than what would be obtained if the mass density were assumed constant (dashed green line). A higher volumetric flow rate means a shorter residence time. Use sliders to change the volumes of the reactor with and without density change in order to make comparisons at the same conversion or the same reactor volume. Use sliders to change the feed reactant concentration, feed volumetric flow rate, and reactor temperature.
, and this causes the mass density to decrease. Thus, the volumetric flow rate increases down the reactor so that the conversion (blue line) is lower than what would be obtained if the mass density were assumed constant (dashed green line). A higher volumetric flow rate means a shorter residence time. Use sliders to change the volumes of the reactor with and without density change in order to make comparisons at the same conversion or the same reactor volume. Use sliders to change the feed reactant concentration, feed volumetric flow rate, and reactor temperature.
Contributed by: Rachael L. Baumann (July 2013)
With additional contributions by: John L. Falconer and Nick Bongiardina
(University of Colorado Boulder, Department of Chemical and Biological Engineering)
Open content licensed under CC BY-NC-SA
Snapshots
Details
Material balances are done to determine the conversion  :
:
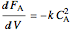 ,
,
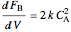 ,
,
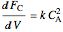 ,
,
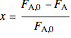 ,
,
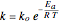 ,
,
where 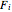 is the molar flow rate of component
is the molar flow rate of component 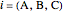 ,
,  is the rate constant,
is the rate constant, 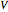 is volume,
is volume, 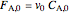 is the feed flow rate of
is the feed flow rate of 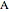 ,
, 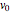 is the constant volumetric flow rate,
is the constant volumetric flow rate, 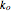 is the pre-exponential factor,
is the pre-exponential factor, 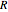 is the ideal gas constant, and
is the ideal gas constant, and 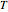 is temperature.
is temperature.
When density change is ignored, the concentration of 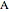
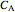 is:
is:
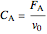 .
.
When density change is taken into account:
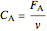 ,
,
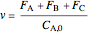 ,
,
where  is volumetric flow rate, which changes with distance down the reactor.
is volumetric flow rate, which changes with distance down the reactor.
The screencast video at [1] show how to use this Demonstration.
Reference
[1] Why Density Change Cannot Be Ignored in a Plug Flow Reactor (PFR). www.colorado.edu/learncheme/kinetics/CantIgnoreDensityPFR.html.
Permanent Citation