Synergism and Antagonism

Requires a Wolfram Notebook System
Interact on desktop, mobile and cloud with the free Wolfram Player or other Wolfram Language products.
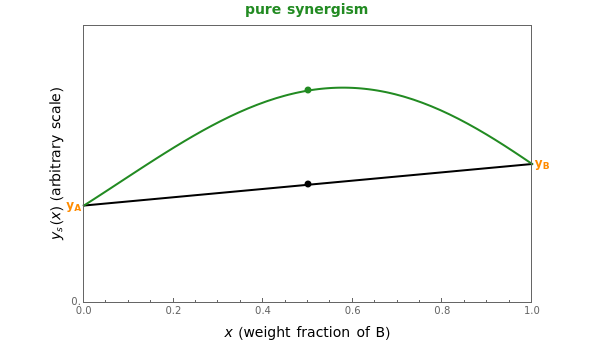
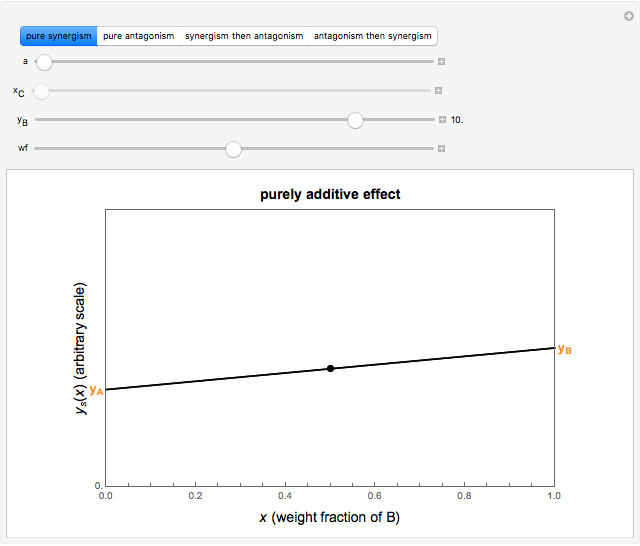
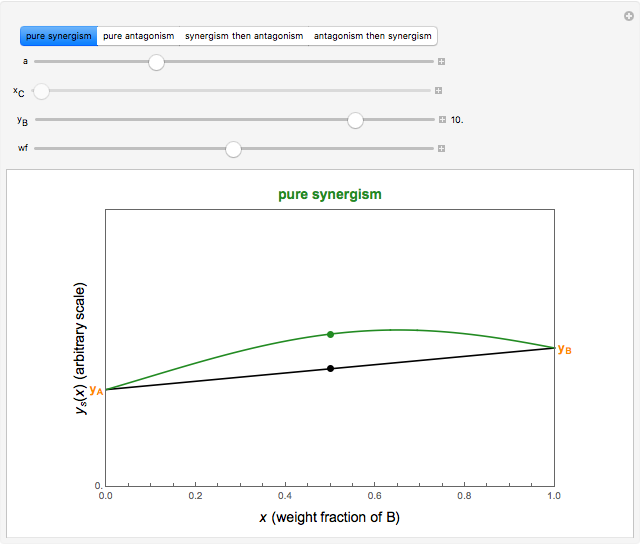
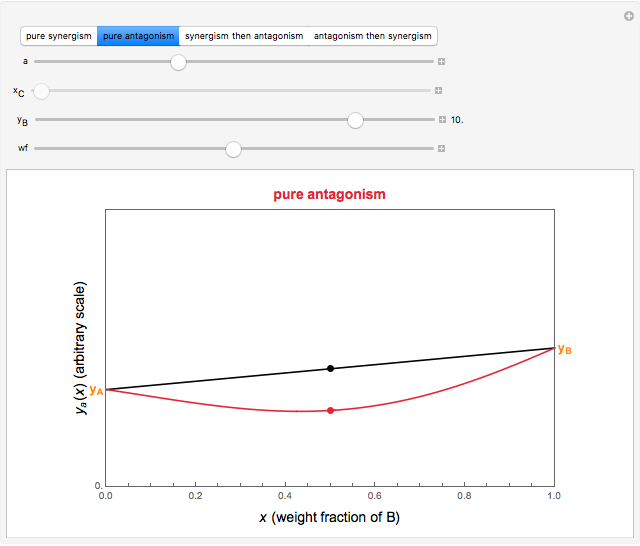
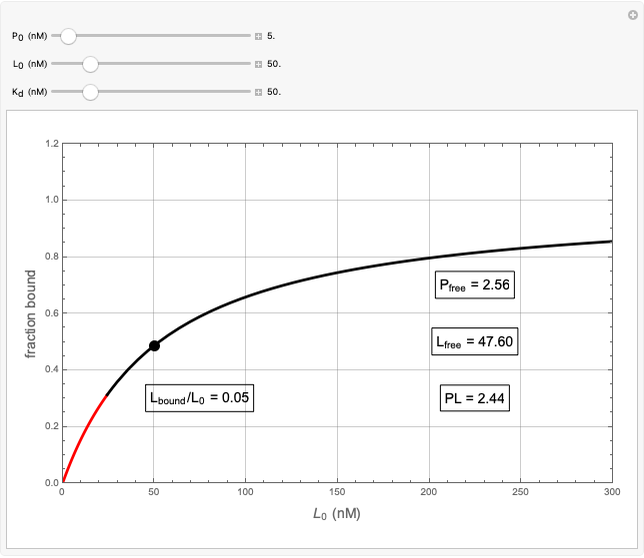
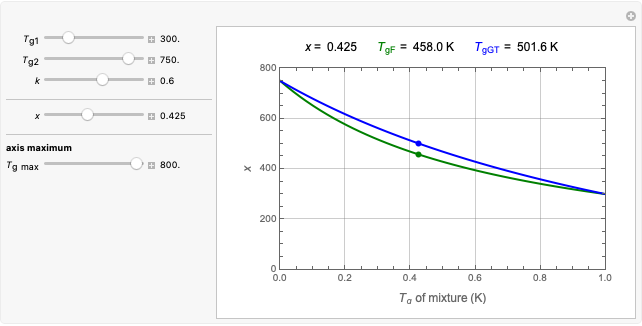
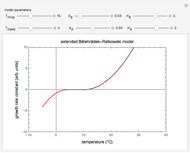
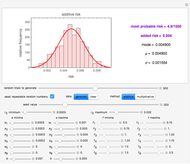
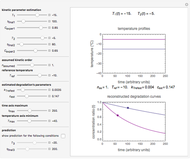
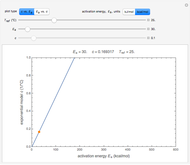
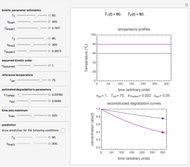
This Demonstration provides visualization of the synergism and antagonism phenomena encountered in the simultaneous application of two antimicrobial agents, drugs, or other biologically active compounds. In synergism, the combined effect is greater than that calculated from the sum of the individual effects, and in antagonism it is smaller. The merely additive effect, due to stoichiometry, is the straight black line connecting the activity of compound A alone with that of compound B alone on the plot of the combined activity versus the mixture's composition. Synergism manifests in a curve, or local part of a curve, lying above this line, and antagonism is below it. The curves generated are schematic and intended solely for illustration; there are four kinds: pure synergism, pure antagonism, synergism turning into antagonism, and antagonism turning into synergism.
Contributed by: Mark D. Normand and Micha Peleg (October 2011)
Open content licensed under CC BY-NC-SA
Snapshots
Details
Snapshot 1: purely additive effect (no synergism or antagonism)
Snapshot 2: pure mild synergism
Snapshot 3: pure mild antagonism
Snapshot 4: pure strong antagonism
Snapshot 5: synergism changing into antagonism
Snapshot 6: antagonism changing into synergism
This Demonstration illustrates graphically the phenomena of synergism and antagonism in hypothetical binary mixtures of two biologically active compounds, A and B. You can generate four principal scenarios by clicking the setter bar. The scenarios are: pure synergism, pure antagonism, synergism turning into antagonism by the rise of compound B's weight fraction  , and antagonism turning into synergism. The range of
, and antagonism turning into synergism. The range of  is from zero (signifying 100% A and 0% B), to one (signifying 0% A and 100% B). What is plotted is an arbitrary curve depicting the mixture's activity or efficacy
is from zero (signifying 100% A and 0% B), to one (signifying 0% A and 100% B). What is plotted is an arbitrary curve depicting the mixture's activity or efficacy  on an arbitrary scale, as a function of
on an arbitrary scale, as a function of  .
.
The activity or efficacy of component A alone is  and that of component B alone is
and that of component B alone is  . Each can be made smaller, greater than, or equal to the other by moving the
. Each can be made smaller, greater than, or equal to the other by moving the  slider. A point on the black line that connects
slider. A point on the black line that connects  and
and  , chosen by moving the "wf" slider, represents a mixture whose effect is merely additive, that is, could be predicted from stoichiometry alone. A curve of pure synergism is above this line for any
, chosen by moving the "wf" slider, represents a mixture whose effect is merely additive, that is, could be predicted from stoichiometry alone. A curve of pure synergism is above this line for any  and a curve of pure antagonism in this range is below it. The intensity of either is represented by the deviation from the straight line between
and a curve of pure antagonism in this range is below it. The intensity of either is represented by the deviation from the straight line between  and
and  in either direction, and can be increased or decreased by moving the "a" slider.
in either direction, and can be increased or decreased by moving the "a" slider.
A transition from synergism to antagonism, or vice versa, is controlled by  , the point where
, the point where  intersects the straight line connecting
intersects the straight line connecting  and
and  , and its location can also be varied by a slider.
, and its location can also be varied by a slider.
The sole purpose of the Demonstration is to provide qualitative images of how the phenomena of synergism and antagonism in binary mixtures are manifested in the general shape of the  versus
versus  relationship. Consequently, we have chosen models of convenience to generate the plots, whose ordinate has arbitrary scale. The same is true for the models' parameters and their ranges. As a result, the
relationship. Consequently, we have chosen models of convenience to generate the plots, whose ordinate has arbitrary scale. The same is true for the models' parameters and their ranges. As a result, the  vs.
vs.  plots do not necessarily match any actual biological system.
plots do not necessarily match any actual biological system.
Although only bioactive materials are mentioned, the Demonstration can also be used to illustrate synergism and antagonism where the effect is physical, as in hydrocolloid interactions that affect their solutions' viscosity [1], or chemical, as when an antioxidant turns into an oxidant or vice versa.
Reference
[1] G. Kaletunc-Gencer and M. Peleg, "Rheological Characteristics of Selected Food Gum Mixtures in Solution," Journal of Texture Studies, 17, 1986 pp. 61–70.
Permanent Citation