Formation of Acetals under Acid Conditions

Requires a Wolfram Notebook System
Interact on desktop, mobile and cloud with the free Wolfram Player or other Wolfram Language products.
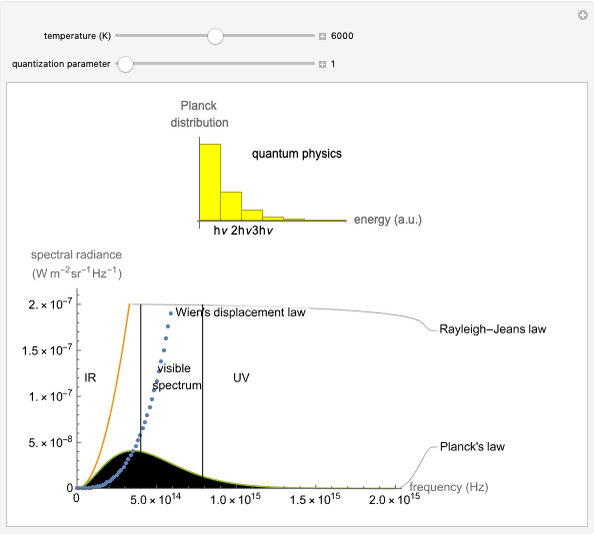
This Demonstration considers the chemical kinetics of the formation of acetals from hemiacetals.
This reaction, unlike the formation of hemiacetals [1], can only take place with acid catalysis. In fact, this reaction is a substitution reaction, replacing the –OH group with the –OR group, and an acid solution is necessary to transform –OH into  , thus obtaining a good leaving group. If an ion is formed and an excess of charge is present, a colored shadow identifies the charge and its center: purple indicates an excess of positive charge, while green indicates an excess of negative charge.
, thus obtaining a good leaving group. If an ion is formed and an excess of charge is present, a colored shadow identifies the charge and its center: purple indicates an excess of positive charge, while green indicates an excess of negative charge.
Contributed by: D. Meliga, V. Giambrone, L. Lavagnino and S. Z. Lavagnino
Additional contribution by: F. Calcagno (I.T.I.S. Artom) (January 2024)
Open content licensed under CC BY-NC-SA
Snapshots
Details
Snapshot 1: first step of the acid catalysis, a proton transfer to start the reaction
Snapshot 2: second step of the acid catalysis, a water molecule loss to form the carbocation
Snapshot 3: third step of the acid catalysis, a nucleophile attack between the carbocation and the methanol
Snapshot 4: final step of the acid catalysis, a proton loss to produce the acetal
References: [1] Addizioni nucleofile ai chetoni https://www.youtube.com/watch?v=dKQLBPJ8Jew&list=PLswwssc6Q2ybl1IVEY56LVc16XsDKzFr-&index=48 [2] Hydrates, Hemiacetals, and Acetals https://www.masterorganicchemistry.com/2010/05/28/acetals-hemiacetals-hydrates/
Permanent Citation