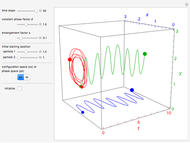
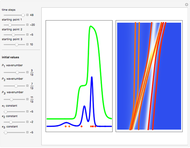
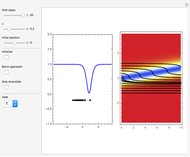
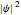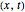The Causal Interpretation of Exciting an Atom by Absorption of Intense Light

Requires a Wolfram Notebook System
Interact on desktop, mobile and cloud with the free Wolfram Player or other Wolfram Language products.
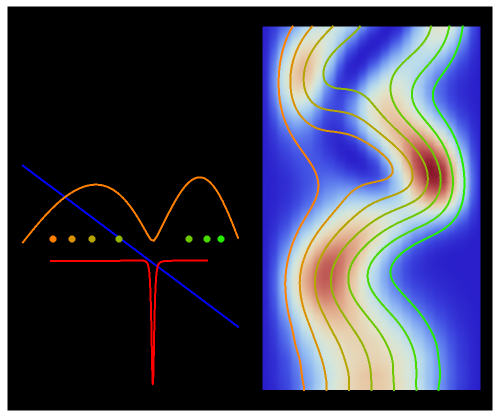
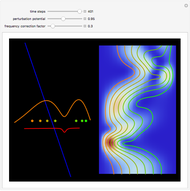
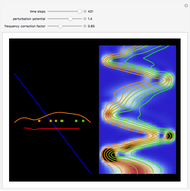
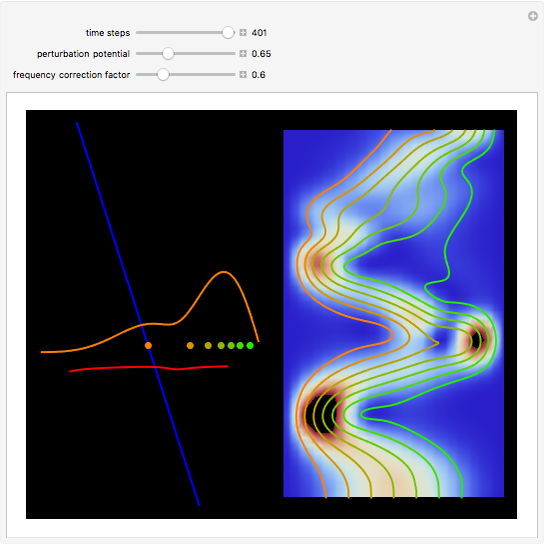
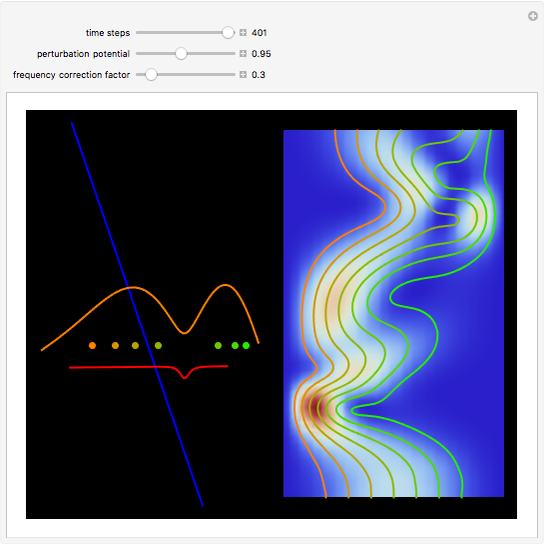
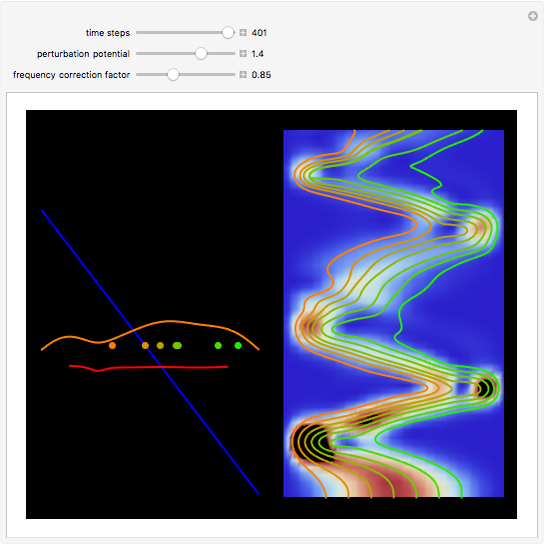
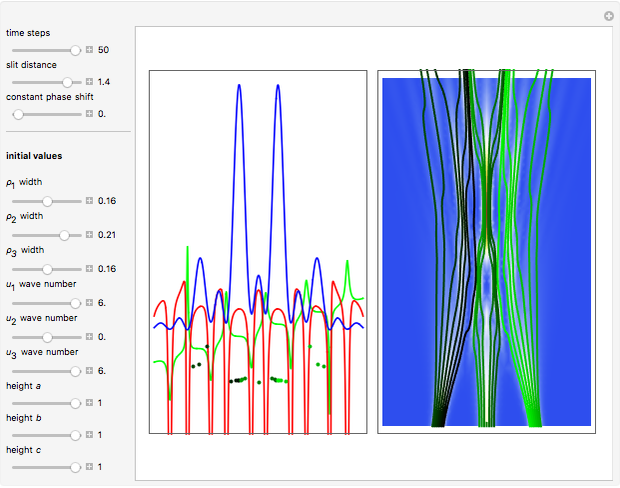
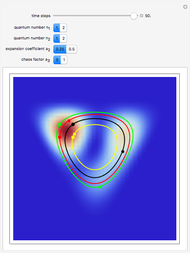
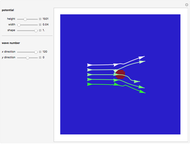
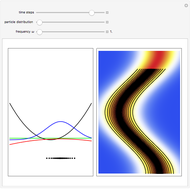
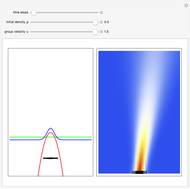
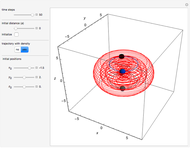
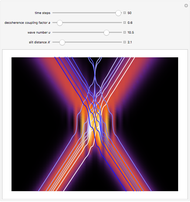
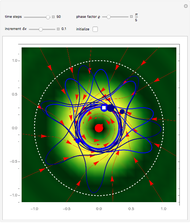
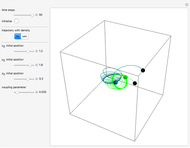
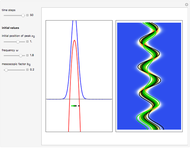
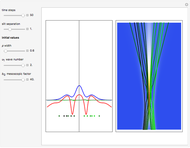
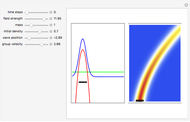
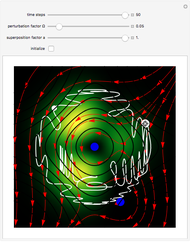
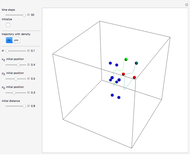
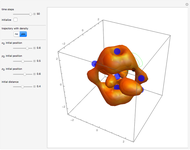
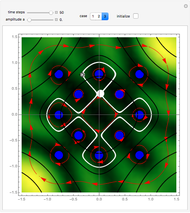
The process of exciting an atom involves adding energy. In the photoelectric effect, electrons are emitted when they absorb energy from incident radiation with a certain minimum frequency  . This Demonstration shows only the energy transition of a perturbed atom. An atom is described by a wave in a box with infinitely high walls. For this particular case, the atom is perturbed by a strong periodic external potential, which could be interpreted as a weak or intense electric field with an electric field strength
. This Demonstration shows only the energy transition of a perturbed atom. An atom is described by a wave in a box with infinitely high walls. For this particular case, the atom is perturbed by a strong periodic external potential, which could be interpreted as a weak or intense electric field with an electric field strength 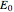 that oscillates with some angular frequency
that oscillates with some angular frequency  . The absorption (or emission) of energy could be described by the transition frequency
. The absorption (or emission) of energy could be described by the transition frequency  of the adjacent quantum energy
of the adjacent quantum energy level
level 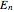 , which is
, which is 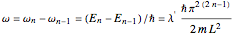 (here, with frequency correction factor
(here, with frequency correction factor 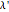 , mass
, mass 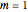 ,
, 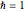 , box length
, box length  , and energy eigenvalue
, and energy eigenvalue  ). Only for definite values of the frequency
). Only for definite values of the frequency  (by a given strength
(by a given strength 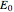 of the perturbation), depending on the energy eigenvalues
of the perturbation), depending on the energy eigenvalues 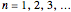 , can the atom undergo a transition from the first to the second excited state. If the perturbation term
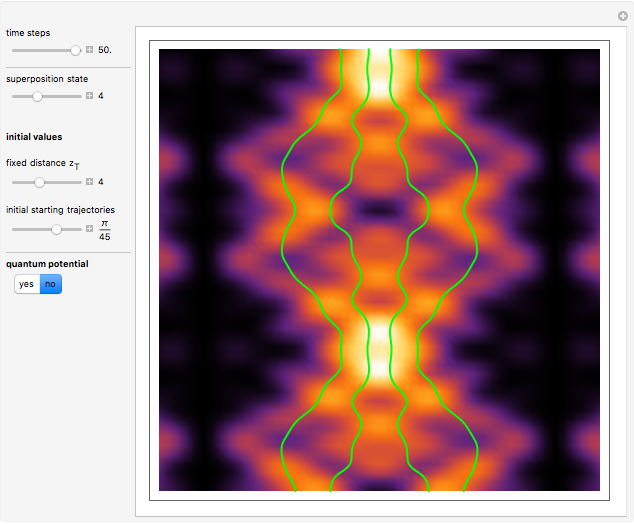
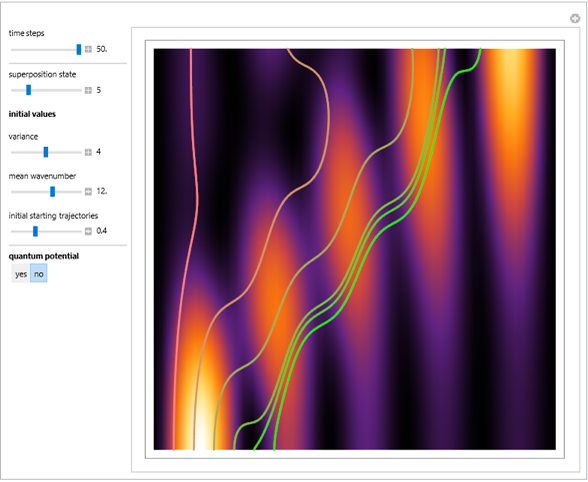
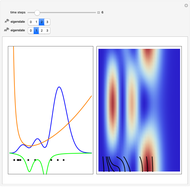
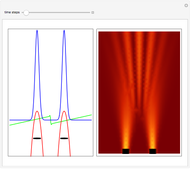
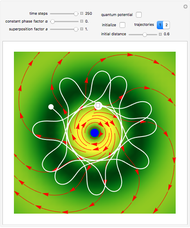
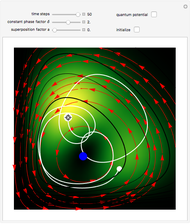
, can the atom undergo a transition from the first to the second excited state. If the perturbation term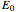 becomes large, the quantum state evolves into a superposition of states with different energy. These mixed states lead to ergodic motion of the quantum particle. The excited atom can be measured by removing the confining well; then the particle moves away faster because of the excited state. The simplicity of the model neglects quantization of the transition-inducing field and other radiation effects.
becomes large, the quantum state evolves into a superposition of states with different energy. These mixed states lead to ergodic motion of the quantum particle. The excited atom can be measured by removing the confining well; then the particle moves away faster because of the excited state. The simplicity of the model neglects quantization of the transition-inducing field and other radiation effects.
Contributed by: Klaus von Bloh (April 2013)
Suggested by: Chris Dewdney
Based on a program by: Enrique Zeleny and Paul Nylander
Open content licensed under CC BY-NC-SA
Snapshots
Details
The numerical methods to calculate the velocity and the quantum potential from a discrete function are, in general, not very stable, but the applied interpolation functions lead to an accurate approximation of the physical event.
For speed, contrary to custom the time steps are increased and the mesh points are decreased, so that the final results have only qualitative character in some cases. Further investigations are needed to find precise parameters for the transition process (for an example, see the YouTube video by the author [1].
The author would like to thank Sören Petrat and the team at the department of mathematics of the Ludwig-Maximilians-University Munich (workgroup Bohmian Mechanics) for their helpful suggestions.
References
[1] K. von Bloh. Quantum Time-Dependent Perturbation Theory of the Infinite Potential Wall. [Video]. (Apr 23, 2013) www.youtube.com/watch?v=q5eQf-aecfA.
[2] C. Dewdney and M. M. Lam, "What Happens During a Quantum Transition?," Information Dynamics (H. Atmanspacher and H. Scheingraber, eds.), New York: Plenum Press, 1991.
[3] S. Goldstein. "Bohmian Mechanics." The Stanford Encyclopedia of Philosophy. (Mar 29, 2013)plato.stanford.edu/entries/qm-bohm.
[4] Bohmian-Mechanics.net. (Mar 29, 2013) www.bohmian-mechanics.net/index.html.
[5] Wikipedia. "Crank-Nicolson Method." (Mar 29, 2013) en.wikipedia.org/wiki/Crank-Nicolson_method.
[6] A. Goldberg, H. Schey, and J. L. Schwartz, "Computer-Generated Motion Pictures of One-Dimensional Quantum-Mechanical Transmission and Reflection Phenomena," American Journal of Physics, 35(3), 1967 pp. 177-186. doi:10.1119/1.1973991.
Permanent Citation